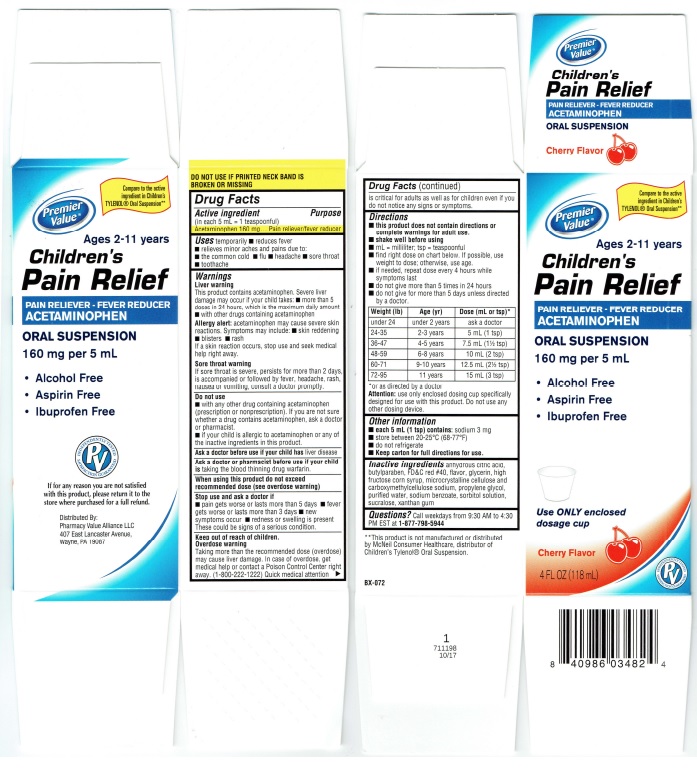
Label: CHILDRENS PAIN RELIEF ORAL SUSPENSION- acetaminophen suspension
- NDC Code(s): 68016-240-04
- Packager: Pharmacy Value Alliance, LLC
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated December 29, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
-
WARNINGS
Warnings
Liver warning
This product contains acetaminophen. Severe liver
damage may occur if your child takes: ■ more than 5
doses in 24 hours, which is the maximum daily amount
■ with other drugs containing acetaminophenAllergy alert: acetaminophen may cause severe skin
reactions. Symptoms may include: ■ skin reddening
■ blisters ■rash
If a skin reaction occurs, stop use and seek medical
help right away.Sore throat warning
If sore throat is severe, persists for more than 2 days,
is accompanied or followed by fever, headache, rash, - DO NOT USE
- ASK DOCTOR
- ASK DOCTOR/PHARMACIST
- WHEN USING
- STOP USE
- Keep out of reach of children
-
OVERDOSAGE
Overdose warning
Taking more than the recommended dose (overdose)
may cause liver damage. In case of overdose, get
medical help or contact a Poison Control Center right
away. (1-800-222-1222) Quick medical attention
is critical for adults as well as for children even if you
do not notice any signs or symptoms. -
DOSAGE & ADMINISTRATION
Directions
■ this product does not contain directions or
complete warnings for adult use.
■ shake well before using
■ mL = milliliter; tsp = teaspoonful
■ find right dose on chart below. If possible, use
weight to dose; otherwise, use age.
■ if needed, repeat dose every 4 hours while
symptoms last
■ do not give more than 5 times in 24 hours
■ do not give for more than 5 days unless directed
by adoctor.Weight (Ib) Age (yr) Dose (mL or tsp)*
under 24 under 2 years ask a doctor
24-35 2-3 years 5 mL (1 tsp)
36-47 4-5 years 7.5 mL (1 Y2 tsp)
48-59 6-8 years 10 mL (2 tsp)
60-71 9-10 year 12.5 mL (2Y2 tsp)
72-95 11 year 15 mL (3 tsp)*or as directed by a doctor
Attention: use only enclosed dosing cup specifically
designed for use with this product. Do not use any
other dosing device. - OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
-
Principal Display Panel
Premier
Value®Children's
Pain ReliefPAIN RELIEVER – FEVER REDUCER
ACETAMINOPHENORAL SUSPENSION
Cherry Flavor
Premier
Value®Compare to the active
ingredients in Children's
TYLENOL® Oral Suspension**Ages 2-11 years
Children's
Pain ReliefPAIN RELIEVER – FEVER REDUCER
ACETAMINOPHENORAL SUSPENSION
160 mg per 5 mL• Alcohol Free
• Aspirin Free
• Ibuprofen FreeUse ONLY enclosed
dosage cupCherry Flavor
4 FL OX (118 mL)INDEPENDENTLY TESTED
PV
SATISFACTION GUARANTEEDDO NOT USE IF PRINTED NECK BAND IS
BROKEN OR MISSING
If for any reason you are not satisfied
with this product, please return it to the
store where purchased for a full refund.Distributed By:
Pharmacy Value Alliance LLC
407 East Lancaster Avenue,
Wayne, PA 19087**This product is not manufactured or distributed
by McNeil Consumer Healthcare, distributor of
Children's Tyleno!® Oral Suspension.BX-072
711198
10/17
Product Label

Premier Value® Children's Pain Relief ACETAMINOPHEN ORAL SUSPENSION Cherry Flavor


end res
-
INGREDIENTS AND APPEARANCE
CHILDRENS PAIN RELIEF ORAL SUSPENSION
acetaminophen suspensionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68016-240 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 160 mg in 5 mL Inactive Ingredients Ingredient Name Strength ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) BUTYLPARABEN (UNII: 3QPI1U3FV8) FD&C RED NO. 40 (UNII: WZB9127XOA) GLYCERIN (UNII: PDC6A3C0OX) HIGH FRUCTOSE CORN SYRUP (UNII: XY6UN3QB6S) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SORBITOL (UNII: 506T60A25R) SUCROSE (UNII: C151H8M554) XANTHAN GUM (UNII: TTV12P4NEE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68016-240-04 1 in 1 CARTON 10/01/2018 1 118 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 10/01/2018 Labeler - Pharmacy Value Alliance, LLC (101668460) Registrant - AptaPharma Inc. (790523323) Establishment Name Address ID/FEI Business Operations AptaPharma Inc. 790523323 manufacture(68016-240)