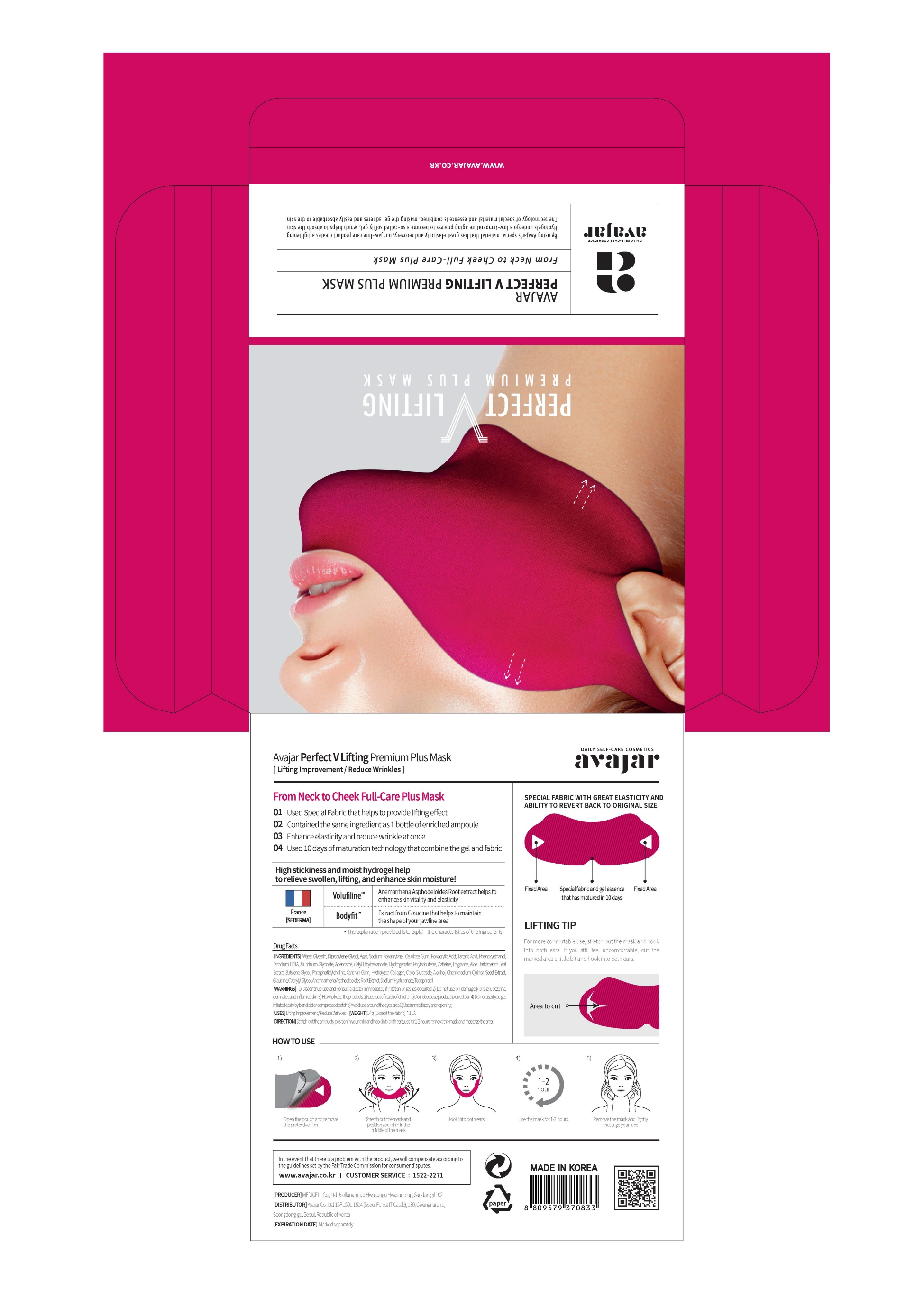
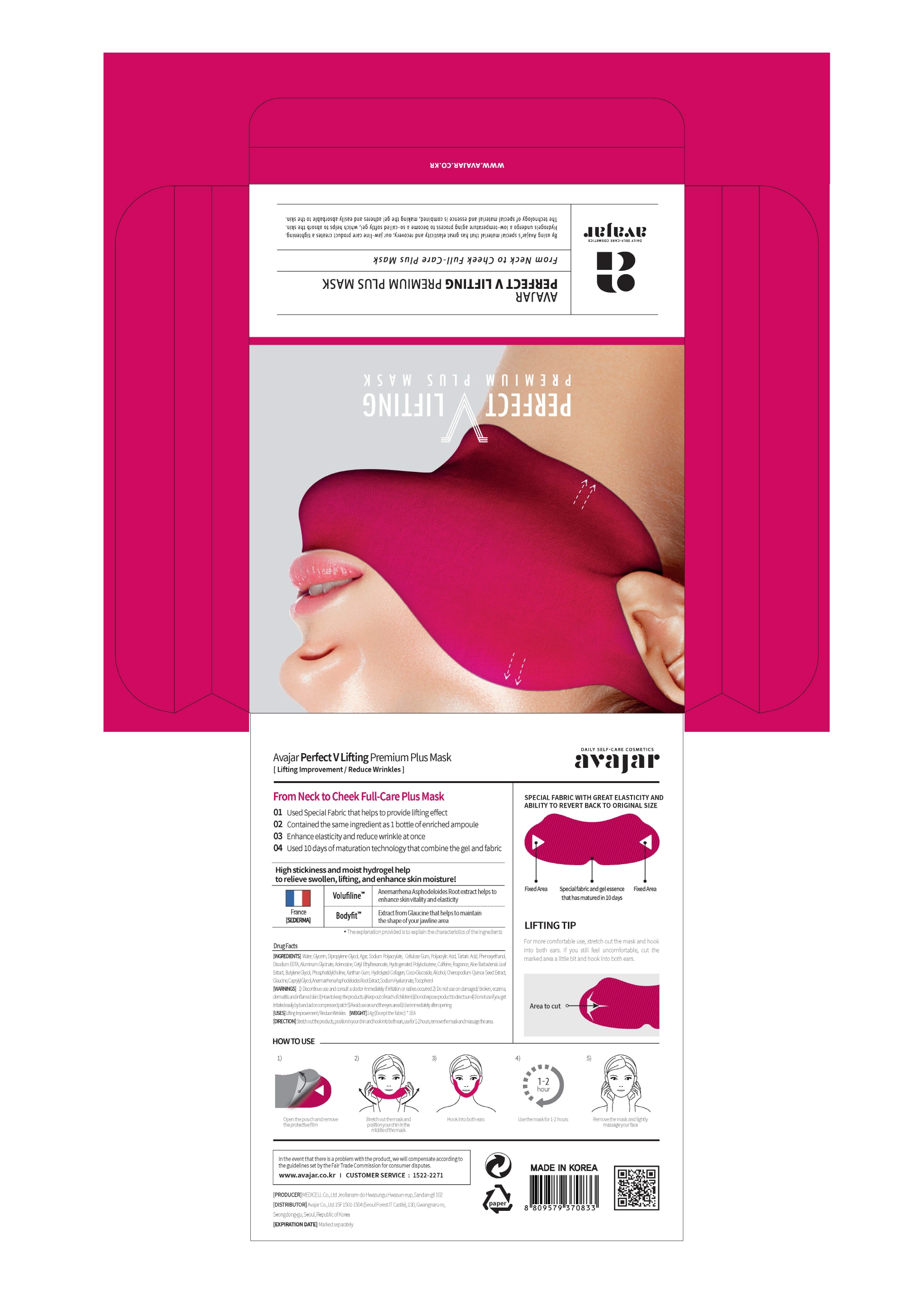
Label: PERFECT V LIFTING PREMIUM PLUS MASK- glycerin liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 71224-0012-1 - Packager: AVAJAR CO LTD
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated May 20, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Water
Dipropylene Glycol
Agar
Sodium Polyacrylate
Cellulose Gum
Polyacrylic Acid
Tartaric acid
Phenoxyethanol
Disodium EDTA
Aluminum Glycinate
Adenosine
Cetyl Ethylhexanoate
Hydrogenated Polyisobutene
Caffeine
Fragrance
Aloe Barbadensis Leaf Extract
Butylene glycol
PHOSPHATIDYLCHOLINE
Xanthan Gum
Hydrolyzed Collagen
Coco-Glucoside
Alcohol
Chenopodium Quinoa Seed Extract
Glaucine
Caprylyl Glycol
Anemarrhena Asphodeloides Root Extract
Sodium Hyaluronate
TOCOPHEROL - PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
- DOSAGE & ADMINISTRATION
-
WARNINGS
1. If the following symptoms occur after product use, stop using the product immediately and consult a dermatologist (continuous use can exacerbate the symptoms).
1) Occurrence of red spots, swelling, itchiness, and other skin irritation
2) If the symptoms above occur after the application area is exposed to direct sunlight
2. Do not use on open wounds, eczema, and other skin irritations
3. Precaution for Storage and Handling
1) Close the lid after use
2) Keep out of reach of infants and children
3) Do not to store in a place with high/low temperature and exposed to direct sunlight - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PERFECT V LIFTING PREMIUM PLUS MASK
glycerin liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71224-0012 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GLYCERIN (UNII: PDC6A3C0OX) (GLYCERIN - UNII:PDC6A3C0OX) GLYCERIN 30 g in 100 g Inactive Ingredients Ingredient Name Strength GLAUCINE (UNII: NU19306XA7) WATER (UNII: 059QF0KO0R) CAFFEINE (UNII: 3G6A5W338E) TARTARIC ACID (UNII: W4888I119H) ADENOSINE (UNII: K72T3FS567) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71224-0012-1 1 in 1 PACKAGE 01/01/2019 1 14 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 01/01/2019 Labeler - AVAJAR CO LTD (689851644) Registrant - AVAJAR CO LTD (689851644) Establishment Name Address ID/FEI Business Operations MEDICELL CO.,LTD 688731040 manufacture(71224-0012) , pack(71224-0012) Establishment Name Address ID/FEI Business Operations AVAJAR CO LTD 689851644 label(71224-0012)