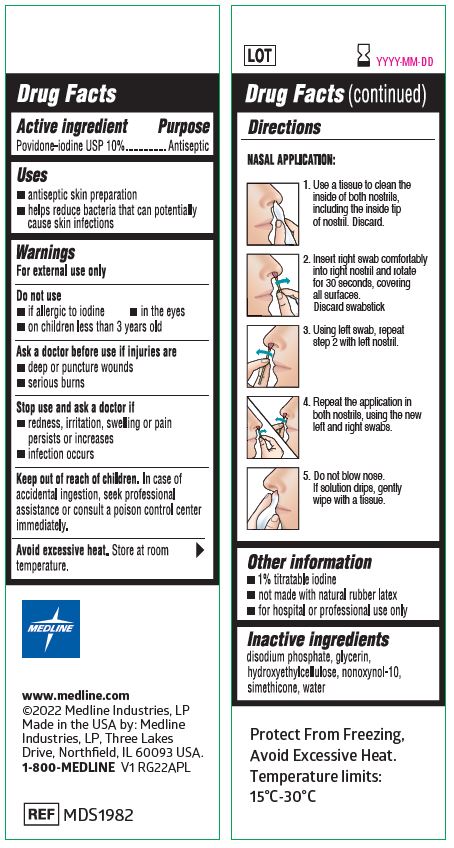
Label: MEDLINE NASAL ANTISEPTIC SWABS- povidone iodine swabstick solution
- NDC Code(s): 53329-497-26, 53329-497-42, 53329-497-66
- Packager: Medline Industries, LP
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated November 30, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
-
Directions
NASAL APPLICATION:

- Use a tissue to clean the inside of both nostrils, including the inside tip of nostril. Discard.
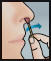
- Insert swab comfortably into one nostril and rotate for 30 seconds, covering all surfaces. Discard swabstick.
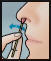
- Using a 2nd swab, repeat step 2 with the other nostril.
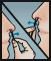
- Repeat the application in both nostrils, using the 3rd and 4th swabs.
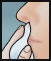
- Do not blow nose. If solution drips, gently wipe with a tissue.
- Other information
- Inactive ingredients
- Manufacturing Information
- Package Label
-
INGREDIENTS AND APPEARANCE
MEDLINE NASAL ANTISEPTIC SWABS
povidone iodine swabstick solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53329-497 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength DIMETHICONE (UNII: 92RU3N3Y1O) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) HYDROXYETHYL CELLULOSE (2000 MPA.S AT 1%) (UNII: S38J6RZN16) NONOXYNOL-10 (UNII: K7O76887AP) GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53329-497-42 2 in 1 BOX 01/15/2018 1 NDC:53329-497-26 10 mL in 1 PACKET; Type 0: Not a Combination Product 2 NDC:53329-497-66 250 in 1 CASE 01/15/2018 2 NDC:53329-497-26 10 mL in 1 PACKET; Type 0: Not a Combination Product 3 NDC:53329-497-26 10 mL in 1 PACKET; Type 0: Not a Combination Product 01/15/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 505G(a)(3) 01/15/2018 Labeler - Medline Industries, LP (025460908) Registrant - Medline Industries, LP (025460908)