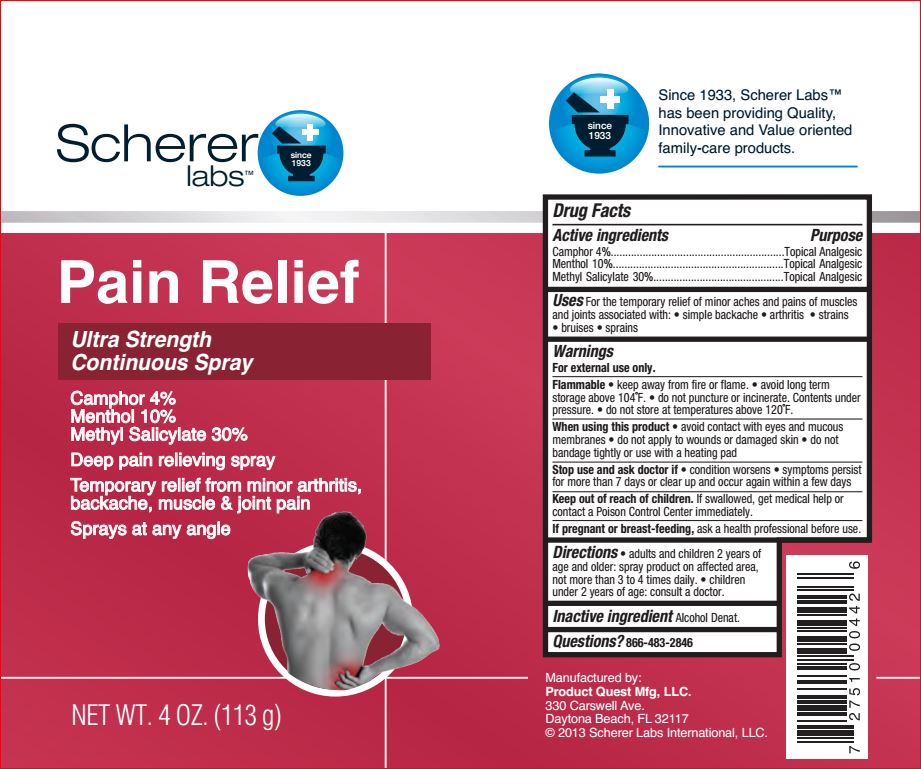
Label: ULTRA STRENGTH PAIN RELIEF- camphor 4%, menthol 10%, methyl salicylate 30% spray
-
Contains inactivated NDC Code(s)
NDC Code(s): 64048-5114-4 - Packager: Product Quest Mfg
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 25, 2018
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients Purpose
- PURPOSE
- Uses
-
Warnings
For external use only
Flammable
• keep away from fire or flame. • avoid long term
storage above 104˚F. • do not puncture or incinerate. Contents under
pressure. • do not store at temperatures above 120˚F• do not use while smoking or near heat or flame
• avoid long term storage above 104F
• do not puncture or incinerate. Contents under pressure
• do not store at temperature above 120F - Stop use and ask a doctor if
- Keep out of reach of the children
- If pregnant or breast-feeding, ask a health professional before use
- Directions
- Inactive ingredients
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ULTRA STRENGTH PAIN RELIEF
camphor 4%, menthol 10%, methyl salicylate 30% sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:64048-5114 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 4 g in 100 g Menthol (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) Menthol 10 g in 100 g Methyl Salicylate (UNII: LAV5U5022Y) (SALICYLIC ACID - UNII:O414PZ4LPZ) Methyl Salicylate 30 g in 100 g Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64048-5114-4 113 g in 1 CAN; Type 0: Not a Combination Product 02/06/2014 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 02/06/2014 Labeler - Product Quest Mfg (927768135) Registrant - Product Quest Mfg (927768135) Establishment Name Address ID/FEI Business Operations Product Quest Mfg 927768135 manufacture(64048-5114) , label(64048-5114)