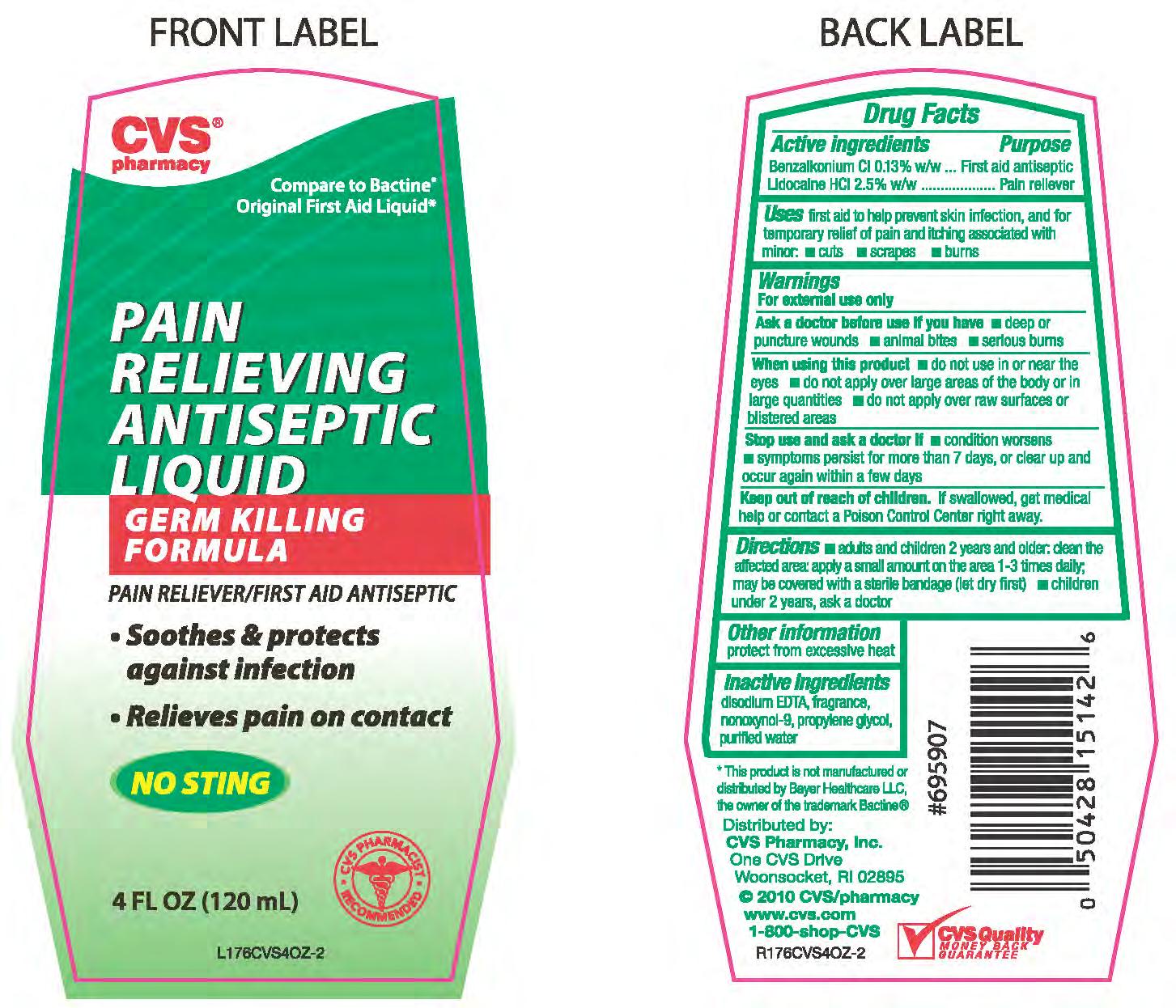
Label: BENZALKONIUM CHLORIDE AND LIDOCAINE HYDROCHLORIDE liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 59779-255-06 - Packager: CVS Pharmacy
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 8, 2010
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
PURPOSE
Active ingredient Purpose
Benzalkonium Cl 0.13% w/w...............................Antiseptic
lidocaine HCl 2.5% w/w......................................Pain reliever
Uses first aid to help prevent skin infection, and for temporary relief of pain
and itching associated with minor - cuts - scrapes - burns
- KEEP OUT OF REACH OF CHILDREN
-
INDICATIONS & USAGE
Directions - adults and children 2 years and older; clean the affected area; apply a
small amount on the area 1-3 times daily; may be covered with a sterile bandage (let dry first)
- children under 2 years, ask a doctor.
Uses first aid to help prevent skin infection, and for temporary relief of pain
and itching associated with minor - cuts - scrapes - burns -
WARNINGS
Warnings
For external use only.
Ask a doctor before use if you have - deep or puncture wounds
- animal bites - serious burns
When using this product - do not use in or near the eyes - do not
apply over large areas of the body or in large quantities - do not apply
over raw surfaces or blistered areas.
Stop use and ask a doctor if - condition worsens - symptoms
persist for more than 7 days, or clear up and occur again within a few days
Keep out of reach of children. If swallowed, get medical
help or contact a Poison Control Center right awayImmediately.
- DOSAGE & ADMINISTRATION
- INACTIVE INGREDIENT
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
BENZALKONIUM CHLORIDE AND LIDOCAINE HYDROCHLORIDE
benzalkonium chloride and lidocaine hydrochloride liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59779-255 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 26 mg in 1 mL LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE 25 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) EDETATE DISODIUM (UNII: 7FLD91C86K) NONOXYNOL-9 (UNII: 48Q180SH9T) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59779-255-06 120 mL in 1 BOTTLE, DISPENSING Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333A 07/08/2010 Labeler - CVS Pharmacy (062312574) Registrant - Pharma Pac, LLC (140807475) Establishment Name Address ID/FEI Business Operations Pharma Pac, LLC 140807475 manufacture