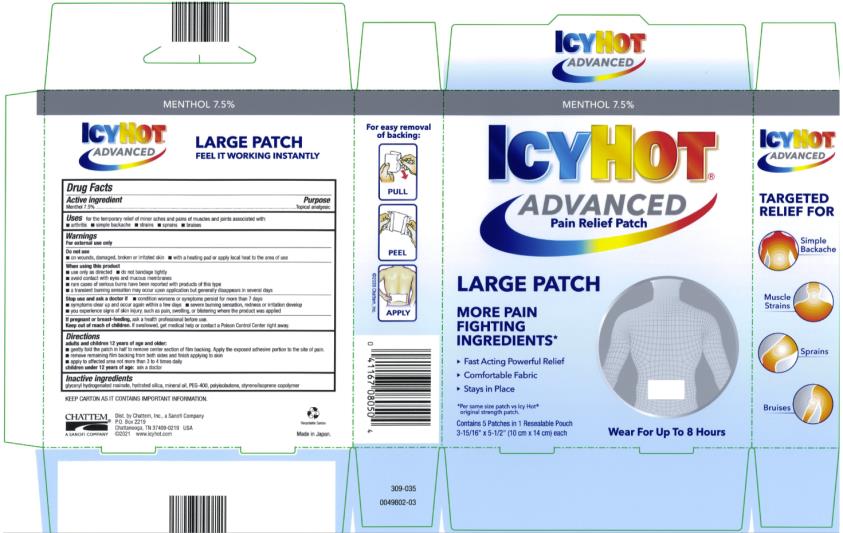
Label: ICY HOT ADVANCED RELIEF- menthol topical analgesic patch
- NDC Code(s): 41167-0805-0, 41167-0805-1
- Packager: Chattem, Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated October 26, 2023
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active Ingredient
- Purpose
- Uses
-
Warnings
For external use only
Do not use
■ on wounds, damaged, broken or irritated skin
■ with a heating pad or apply local heat to the area of use
When using this product
■ use only as directed
■ do not bandage tightly
■ avoid contact with eyes and mucous membranes
■ rare cases of serious burns have been reported with products of this type
■ a transient burning sensation may occur upon application but generally disappears in several days
Stop use and ask a doctor if
■ condition worsens or symptoms persist for more than 7 days
■ symptoms clear up and occur again within a few days
■ severe burning sensation, redness or irritation develop
■ you experience signs of skin injury, such as pain, swelling, or blistering where the product was applied
-
Directions
adults and children 12 years of age and older:
■ gently fold the patch in half to remove center section of film backing. Apply the exposed adhesive portion to the site of pain.
■ remove remaining film backing from both sides and finish applying to skin
■ apply to affected area not more than 3 to 4 times daily
children under 12 years of age: consult a doctor
- Inactive ingredients
- Principal Display Panel
-
INGREDIENTS AND APPEARANCE
ICY HOT ADVANCED RELIEF
menthol topical analgesic patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:41167-0805 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 210 mg Inactive Ingredients Ingredient Name Strength HYDRATED SILICA (UNII: Y6O7T4G8P9) MINERAL OIL (UNII: T5L8T28FGP) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POLYISOBUTYLENE (1000 MW) (UNII: 5XB3A63Y52) GLYCEROL ESTER OF HYDROGENATED ROSIN (UNII: XH62RP786N) STYRENE/ISOPRENE/STYRENE BLOCK COPOLYMER (UNII: K7S96QM8DV) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:41167-0805-0 1 in 1 CARTON 12/01/2012 03/31/2017 1 4 in 1 POUCH; Type 0: Not a Combination Product 2 NDC:41167-0805-1 5 in 1 CARTON; Type 0: Not a Combination Product 10/22/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 12/01/2012 Labeler - Chattem, Inc. (003336013)