Label: BZK TOWELETTE- benzalkonium chloride swab
- NDC Code(s): 65517-0004-1, 65517-0004-2
- Packager: Dukal LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 21, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
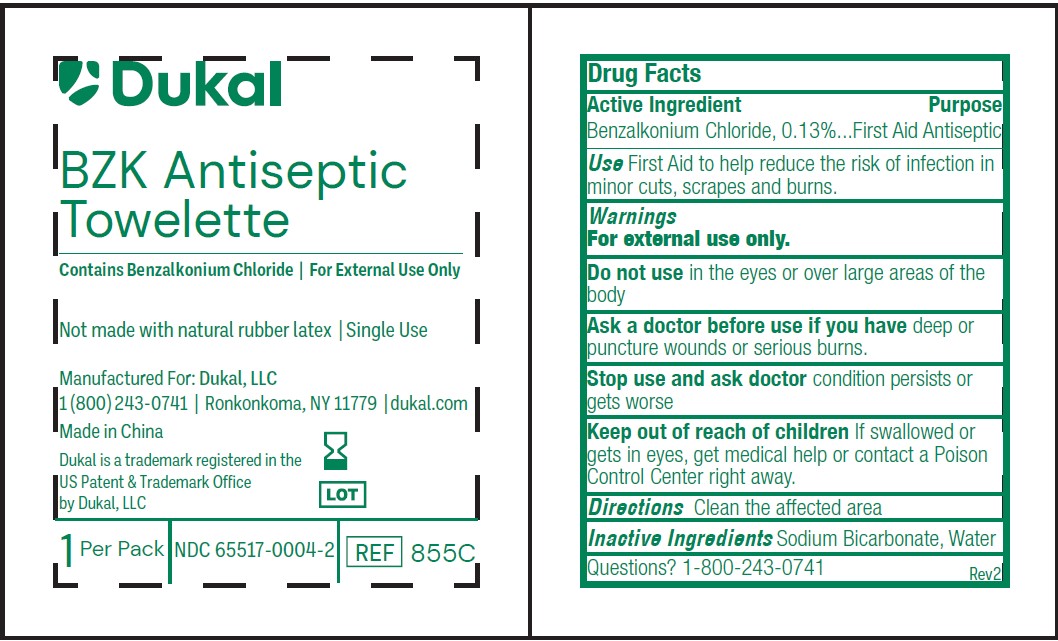
- Active Ingredients
- Use
- Purpose
- Do Not Use
- Stop Use
- Caution
- Inactive Ingredients
-
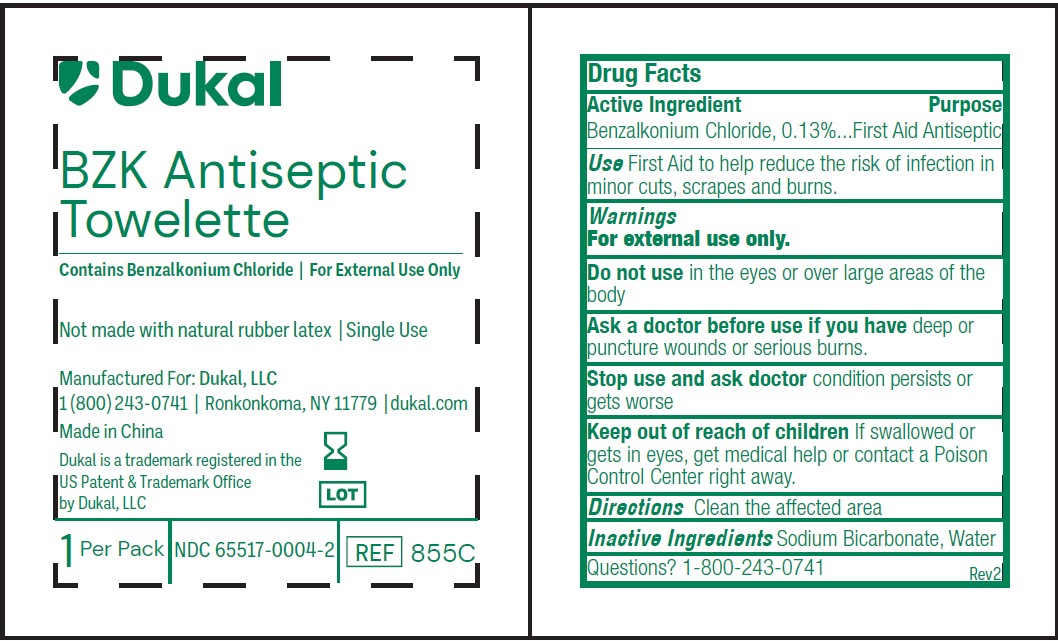
Principal Display Panel - 1.4 mL Pouch Label
Dukal
BZK ANTISEPTICTOWELETTE
Contains Benzalkonium Chloride / For External Use Only
Not made with natural rubber latex / Single Use
Manufactured For: Dukal, LLC
1(800)243-0741 / Ronkonkoma, NY 11779 / dukal.com
Made in China
Dukal is a trademark registered in the
US Patent & Trademark Office
by Dukal, LLC
1 Per Pack / NDC 65517-0004-1 REF 855

-
Principal Display Panel - 2.1 mL Pouch Label
Dukal
BZK ANTISEPTICTOWELETTE
Contains Benzalkonium Chloride / For External Use Only
Not made with natural rubber latex / Single Use
Manufactured For: Dukal, LLC
1(800)243-0741 / Ronkonkoma, NY 11779 / dukal.com
Made in China
Dukal is a trademark registered in the
US Patent & Trademark Office
by Dukal, LLC
1 Per Pack / NDC 65517-0004-2 / REF 855C
-
INGREDIENTS AND APPEARANCE
BZK TOWELETTE
benzalkonium chloride swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:65517-0004 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 1.3 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65517-0004-1 1.4 mL in 1 POUCH; Type 0: Not a Combination Product 01/01/2006 2 NDC:65517-0004-2 2.1 mL in 1 POUCH; Type 0: Not a Combination Product 01/01/2006 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 01/01/2006 Labeler - Dukal LLC (791014871)