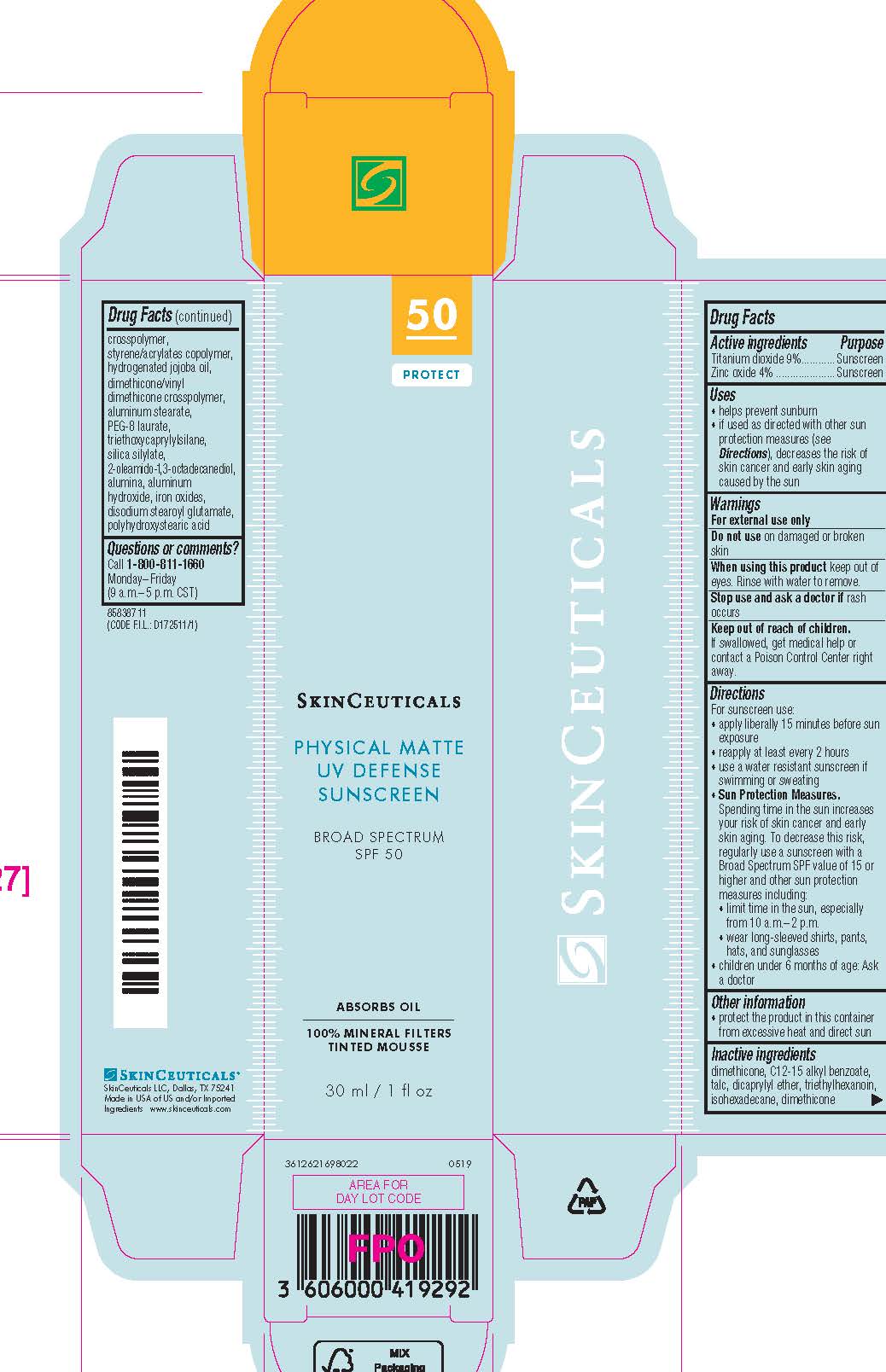
Label: SKINCEUTICALS PHYSICAL MATTE UV DEFENSE BROAD SPECTRUM SPF 50 SUNSCREEN- titanium dioxide and zinc oxide lotion
- NDC Code(s): 49967-929-01, 49967-929-02
- Packager: L'Oreal USA Products Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Uses
- Warnings
- Do not use
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children.
-
Directions
● apply liberally 15 minutes before sun exposure
● reapply:
● after 80 minutes of swimming or sweating
● immediately after towel drying
● at least every 2 hours
● Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
● limit time in the sun, especially from 10 a.m. – 2 p.m.
● wear long-sleeved shirts, pants, hats, and sunglasses
● children under 6 months of age: Ask a doctor
- Other information
-
Inactive ingredients
dimethicone, C12-15 alkyl benzoate, dicaprylyl ether, dimethicone/vinyl dimethicone crosspolymer, talc, triethylhexanoin, isohexadecane, styrene/acrylates copolymer, hydrogenated jojoba oil, aluminum hydroxide, stearic acid, aluminum stearate, iron oxides, silica silylate, alumina, polyhydroxystearic acid, PEG-8 laureth, tocopherol, water, diethylhexyl syringylidenemalonate, laureth-4, disodium stearoyul glutamate, cassia alata leaf extract, maltodextrin, caprylic/capric triglyceride
- Questions or comments?
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
SKINCEUTICALS PHYSICAL MATTE UV DEFENSE BROAD SPECTRUM SPF 50 SUNSCREEN
titanium dioxide and zinc oxide lotionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:49967-929 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 90 mg in 1 mL ZINC OXIDE (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 40 mg in 1 mL Inactive Ingredients Ingredient Name Strength DIMETHICONE (UNII: 92RU3N3Y1O) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) TALC (UNII: 7SEV7J4R1U) DICAPRYLYL ETHER (UNII: 77JZM5516Z) TRIETHYLHEXANOIN (UNII: 7K3W1BIU6K) ISOHEXADECANE (UNII: 918X1OUF1E) DIMETHICONE CROSSPOLYMER (450000 MPA.S AT 12% IN CYCLOPENTASILOXANE) (UNII: UF7620L1W6) STYRENE/ACRYLAMIDE COPOLYMER (500000 MW) (UNII: 5Z4DPO246A) HYDROGENATED JOJOBA OIL (UNII: 7F674YQ5SO) DIMETHICONE/VINYL DIMETHICONE CROSSPOLYMER (HARD PARTICLE) (UNII: H895X08VNQ) ALUMINUM STEARATE (UNII: U6XF9NP8HM) PEG-8 LAURATE (UNII: 762O8IWA10) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) 2-OLEAMIDO-1,3,4-OCTADECATRIYL PROLINATE (UNII: 690VHX6497) ALUMINUM OXIDE (UNII: LMI26O6933) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) FERRIC OXIDE RED (UNII: 1K09F3G675) DISODIUM STEAROYL GLUTAMATE (UNII: 45ASM2L11M) POLYHYDROXYSTEARIC ACID (2300 MW) (UNII: YXH47AOU0F) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:49967-929-01 1 in 1 CARTON 03/01/2015 1 30 mL in 1 TUBE; Type 0: Not a Combination Product 2 NDC:49967-929-02 1 in 1 CARTON 03/01/2015 2 4 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 03/01/2015 Labeler - L'Oreal USA Products Inc (002136794) Establishment Name Address ID/FEI Business Operations Goodier Cosmetics LP 007317209 manufacture(49967-929)