Label: QC QUALITY CHOICE PINK BISMUTH STOMACH RELIEF- bismuth subsalicylate suspension
-
Contains inactivated NDC Code(s)
NDC Code(s): 63868-303-08 - Packager: Chain Drug Manufacturing Assn
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 18, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
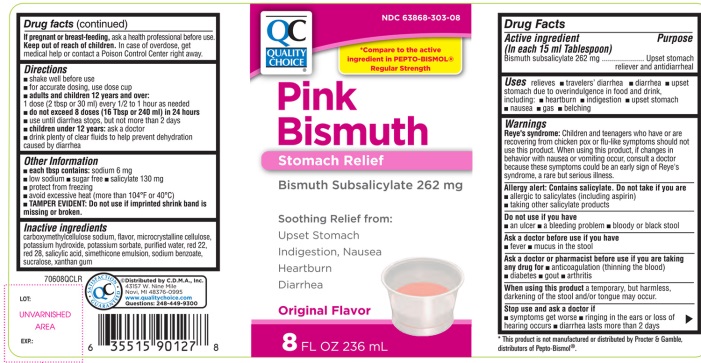
- Active ingredient(in each 15 ml Tablespoon)
- Purposes
- Uses
-
Warnings
Reye's syndrome:
Children and teenagers who have or are recovering from chicken pox or flu-like symptoms should not use this product. When using this product, if changes in behavior with nausea and vomiting occur, consult a doctor because these symptoms could be an early sign of Reye's syndrome, a rare but serious illness.
Allergy alert: Contains salicylate. Do not take if you are
- ▪
- allergic to salicylates (including aspirin)
- ▪
- taking other salicylate products
Ask a doctor or pharmacist before use if you are taking any drug for
- ▪
- anticoagulation (thinning the blood)
- ▪
- diabetes
- ▪
- gout
- ▪
- arthritis
-
Directions
- ▪
- shake well before use
- ▪
- for accurate dosing, use dose cup
- ▪
-
adults and children 12 years and over:
1 dose (2 tbsp or 30 ml) every ½ to 1 hour as needed - ▪
- do not exceed 8 doses (16 tbsp or 240 ml) in 24 hours
- ▪
- use until diarrhea stops but not more than 2 days
- ▪
- children under 12 years: ask a doctor
- ▪
- drink plenty of clear fluids to help prevent dehydration caused by diarrhea
- Other information
- Inactive ingredients
-
PRINCIPAL DISPLAY PANEL - 236 mL Bottle Label
QC
Quality Choice®
NDC 63868-303-08
*Compare to the active ingredient in PEPTO-BISMOL® Regular strength
PINK BISMUTH
Stomach Relief
Bismuth Subsalicylate 262mg
Soothing Relief from:
- Upset Stomach
- Indigestion, Nausea
- Heartburn
- •
- Diarrhea
- NaN
- Original Flavor
8 FL OZ (236 mL)
QC SATISFACTION GUARANTEED
©Distributed by C.D.M.A., Inc.
43157 W. Nine Mile
Novi, MI 48376-0995
Questions: 248-449-9300
*This product is not manufactured or distributed by Procter & Gamble, distributors of Pepto-Bismol®.
-
INGREDIENTS AND APPEARANCE
QC QUALITY CHOICE PINK BISMUTH STOMACH RELIEF
bismuth subsalicylate suspensionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63868-303 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Bismuth subsalicylate (UNII: 62TEY51RR1) (Salicylic acid - UNII:O414PZ4LPZ, Bismuth cation - UNII:ZS9CD1I8YE) Bismuth subsalicylate 262 mg in 15 mL Inactive Ingredients Ingredient Name Strength CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) POTASSIUM HYDROXIDE (UNII: WZH3C48M4T) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) WATER (UNII: 059QF0KO0R) D&C red no. 22 (UNII: 1678RKX8RT) D&C red no. 28 (UNII: 767IP0Y5NH) salicylic acid (UNII: O414PZ4LPZ) DIMETHICONE (UNII: 92RU3N3Y1O) SODIUM BENZOATE (UNII: OJ245FE5EU) SUCRALOSE (UNII: 96K6UQ3ZD4) XANTHAN GUM (UNII: TTV12P4NEE) Product Characteristics Color PINK Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63868-303-08 236 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 03/23/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part335 03/23/2015 Labeler - Chain Drug Manufacturing Assn (011920774)