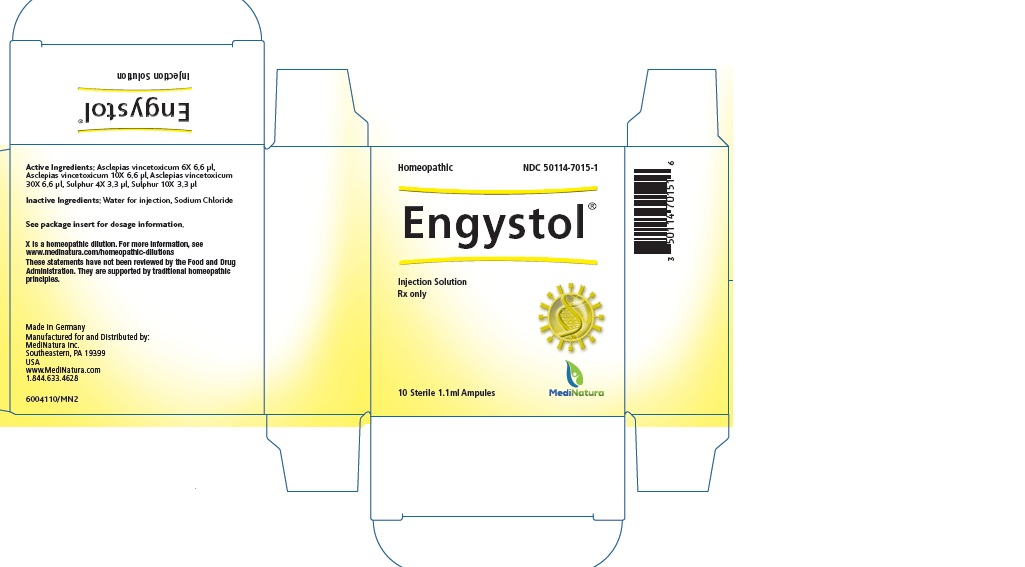
Label: ENGYSTOL- cynanchum vincetoxicum root and sulfur injection
- NDC Code(s): 50114-7015-1, 50114-7015-2
- Packager: MediNatura
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated October 14, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DESCRIPTION
- INDICATIONS AND USAGE
-
DOSAGE AND ADMINISTRATION
General Considerations
• The dosage schedules listed below can be used as a general guide for the administration of Engystol® Injection Solution.
• Engystol® Injection Solution may be administered s.c., i.d., i.m., or i.v.
• The interval between injections is left to the discretion of the HCP, but should not exceed 1 ampule in 24 hours.
• Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard any unused ampule contents.
• Draw up required dose into syringe.
• Discard any unused ampule contents. Do not reuse ampule.
-
Only licensed practitioners with sufficient expertise in injecting drugs, including the respective route of administration, should administer the product.
Standard Dosage:
Adults and children 12 years and older: 1 ml 1 to 3 times per 7 days.
Children 6 to 11 years: 0.7 ml 1 to 3 times per 7 days.
Children 2 to 5 years: 0.5 ml 1 to 3 times per 7 days.
Acute Dosage:
Adults and children 12 years and older: 1 ml daily, and then continue with standard dosage.
Children 6 to 11 years: 0.7 ml daily, and then continue with standard dosage.
Children 2 to 5 years: 0.5 ml daily, and then continue with standard dosage.
-
- CONTRAINDICATIONS
- WARNINGS AND PRECAUTIONS
-
ADVERSE REACTIONS
Post-marketing Experience
• The following adverse events have been identified during post-marketing use of Engystol® Injection Solution. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
• Allergic (hypersensitivity) skin reactions may occur in isolated cases.
To report SUSPECTED ADVERSE REACTIONS, contact MediNatura. at 1.844.633.4628 or info@medinatura.com or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch
- OVERDOSAGE
- CLINICAL PHARMACOLOGY
- DOSAGE
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ENGYSTOL
cynanchum vincetoxicum root and sulfur injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:50114-7015 Route of Administration INTRADERMAL, INTRAMUSCULAR, INTRAVENOUS, SUBCUTANEOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CYNANCHUM VINCETOXICUM ROOT (UNII: 9R858U917W) (CYNANCHUM VINCETOXICUM ROOT - UNII:9R858U917W) CYNANCHUM VINCETOXICUM ROOT 6 [hp_X] in 1.1 mL SULFUR (UNII: 70FD1KFU70) (SULFUR - UNII:70FD1KFU70) SULFUR 4 [hp_X] in 1.1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50114-7015-1 10 in 1 CARTON 05/31/2014 02/28/2025 1 1.1 mL in 1 AMPULE; Type 0: Not a Combination Product 2 NDC:50114-7015-2 3 in 1 CARTON 05/31/2014 02/28/2025 2 1.1 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 05/31/2014 02/28/2025 Labeler - MediNatura (102783016) Establishment Name Address ID/FEI Business Operations Hameln Pharma GmbH 315869123 manufacture(50114-7015) Establishment Name Address ID/FEI Business Operations Biologische Heilmittel Heel 315635359 manufacture(50114-7015)