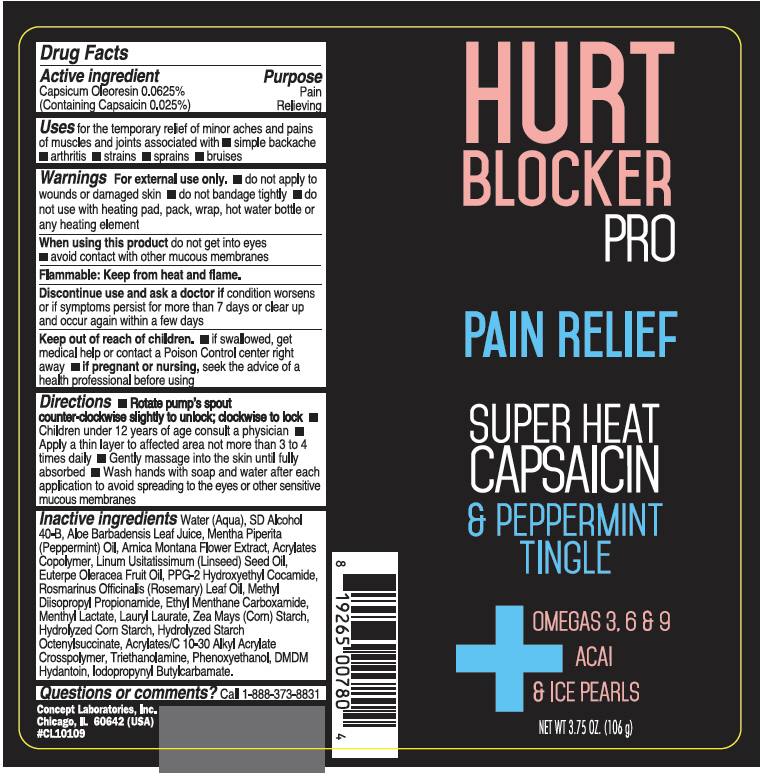
Label: HURT BLOCKER PRO PAIN RELIEF- capsaicin cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 64058-246-42, 64058-246-64 - Packager: Concept Laboratories, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated June 23, 2015
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only.
- do not apply to wounds or damaged skin
- do not bandage tightly
- do not use with heating pad, pack, wrap, hot water bottle or any heating element
When using this product do not get into eyes
- avoid contact with other mucous membranes
Flammable: Keep from heat and flame.
-
Directions
- Rotate pump's spout counter-clockwise slightly to unlock; clockwise to lock
- Children under 12 years of age consult a physician
- Apply a thin layer to affected area not more than 3 to 4 times daily
- Gently massage into the skin until fully absorbed
- Wash hands with soap and water after each application to avoid spreading to the eyes or other sensitive mucous membranes
-
Inactive ingredients
Water (Aqua), SD Alcohol 40-B, Aloe Barbadensis Leaf Juice, Mentha Piperita (Peppermint) Oil, Arnica Montana Flower Extract, Acrylates Copolymer, Linum Usitatissimum (Linseed) Seed Oil, Euterpe Oleracea Fruit Oil, PPG-2 Hydroxyethyl Cocamide, Rosmarinus Officinalis (Rosemary) Leaf Oil, Methyl Diisopropyl Propionamide, Ethyl Menthane Carboxamide, Menthyl Lactate, Lauryl Laurate, Zea Mays (Corn) Starch, Hydrolyzed Corn Starch, Hydrolyzed Starch Octenylsuccinate, Acrylates/C 10-30 Alkyl Acrylate Crosspolymer, Triethanolamine, Phenoxyethanol, DMDM Hydantoin, Iodopropynyl Butylcarbamate.
- Questions or comments?
- PRINCIPAL DISPLAY PANEL - 106 g Bottle Label
-
INGREDIENTS AND APPEARANCE
HURT BLOCKER PRO PAIN RELIEF
capsaicin creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:64058-246 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Capsaicin (UNII: S07O44R1ZM) (Capsaicin - UNII:S07O44R1ZM) Capsaicin 0.25 mg in 1 g Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) ARNICA MONTANA FLOWER (UNII: OZ0E5Y15PZ) METHYL DIISOPROPYL PROPIONAMIDE (UNII: 6QOP5A9489) LAURYL LAURATE (UNII: GPW77G0937) PHENOXYETHANOL (UNII: HIE492ZZ3T) DMDM HYDANTOIN (UNII: BYR0546TOW) IODOPROPYNYL BUTYLCARBAMATE (UNII: 603P14DHEB) ALOE VERA LEAF (UNII: ZY81Z83H0X) MENTHYL LACTATE, (-)- (UNII: 2BF9E65L7I) STARCH, CORN (UNII: O8232NY3SJ) PPG-2 HYDROXYETHYL COCAMIDE (UNII: 34N07GUJ3X) TROLAMINE (UNII: 9O3K93S3TK) ALCOHOL (UNII: 3K9958V90M) PEPPERMINT OIL (UNII: AV092KU4JH) LINSEED OIL (UNII: 84XB4DV00W) ACAI OIL (UNII: Z0W6766A2W) ETHYL MENTHANE CARBOXAMIDE (UNII: 6S7S02945H) ROSEMARY OIL (UNII: 8LGU7VM393) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:64058-246-42 57 g in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product 2 NDC:64058-246-64 106 g in 1 BOTTLE, PUMP; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC MONOGRAPH NOT FINAL part348 05/12/2015 Labeler - Concept Laboratories, Inc. (962282612) Establishment Name Address ID/FEI Business Operations Concept Laboratories, Inc. 962282612 MANUFACTURE(64058-246)