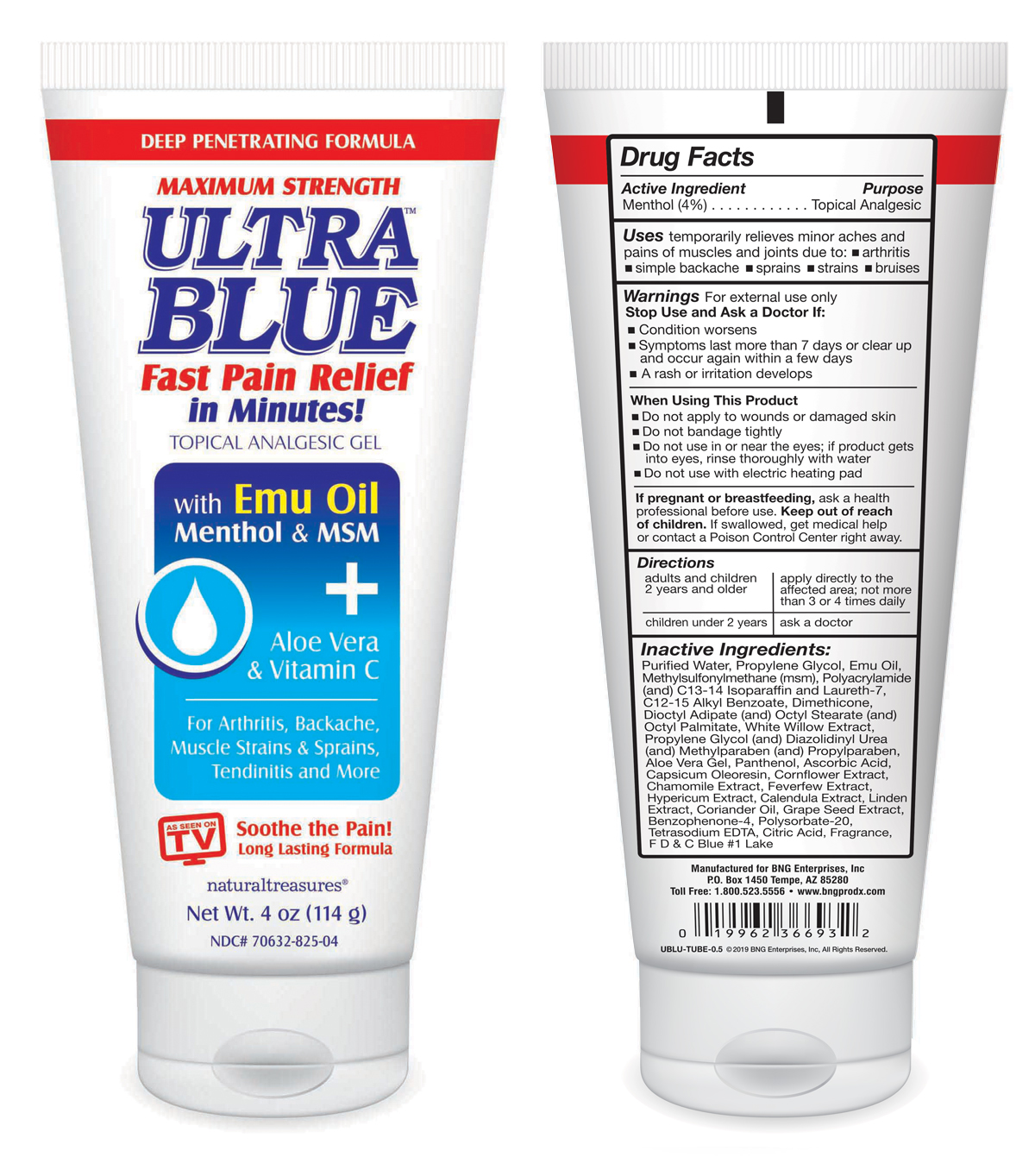
Label: ULTRA BLUE- 4% menthol gel
-
Contains inactivated NDC Code(s)
NDC Code(s): 70632-825-04 - Packager: BNG Enterprises DBA Herbal Clean
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 19, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient section
- Purpose
- Uses:
- Warnings
- Stop Use and Ask a Doctor If
- Keep out of reach of children
- When Using This Product
- Directions
-
Inactive Ingredients:
Purified Water, Propylene Glycol, Emu Oil, Methylsulfonylmethane (msm), Polyacrylamide (and) C13-14 Isoparaffin and Laureth-7, C12-15 Alkyl Benzoate, Dimethicone, Dioctyl Adipate (and) Octyl Stearate (and) Octyl Palmitate, White Willow Extract, Propylene Glycol (and) Diazolidinyl Urea (and) Methylparaben (and) Propylparaben, Aloe Vera Gel, Panthenol, Ascorbic Acid, Capsicum Oleoresin, Cornflower Extract, Chamomile Extract, Feverfew Extract, Hypericum Extract, Calendula Extract, Linden Extract, Coriander Oil, Grape Seed Extract, Benzophenone-4, Polysorbate-20, Tetrasodium EDTA, Citric Acid, Fragrance, F D & C Blue #1 Lake
- Ultra Blue tube front panel, back panel
-
INGREDIENTS AND APPEARANCE
ULTRA BLUE
4% menthol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:70632-825 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) (MENTHOL, UNSPECIFIED FORM - UNII:L7T10EIP3A) MENTHOL, UNSPECIFIED FORM 4.56 g in 114 g Inactive Ingredients Ingredient Name Strength EMU OIL (UNII: 344821WD61) Product Characteristics Color blue Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70632-825-04 5000 g in 1 TUBE; Type 0: Not a Combination Product 07/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 07/01/2018 Labeler - BNG Enterprises DBA Herbal Clean (781993902) Establishment Name Address ID/FEI Business Operations Personal Care Performance Group 144297160 manufacture(70632-825)