Label: EQUI-PHAR- phenylbutazone injection
- NDC Code(s): 50989-425-12
- Packager: Vedco, Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Animal Drug Application
Drug Label Information
Updated May 2, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- GENERAL PRECAUTIONS
-
DESCRIPTION
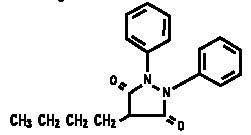
DESCRIPTION: Phenylbutazone 20% Injection (phenylbutazone) is a synthetic, nonhormonal anti-inflammatory, antipyretic compound useful in the management of inflammatory conditions. The apparent analgesic effect is probably related mainly to the compound's anti-inflammatory properties. Chemically, phenylbutazone is 4-butyl-1,2-diphenyl-3,5-pyrazolidinedione. It is a pyrazolon derivative entirely unrelated to the steroid hormones, and has the following structural formula:
-
BACKGROUND PHARMACOLOGY
Kuzell,1,2,3
Payne,4 Fleming,5 and Denko6 demonstrated
clinical effectiveness of phenylbutazone in acute
rheumatism, gout, gouty arthritis, and various
other rheumatoid disorders in man. Anti-rheumatic
and anti-inflammatory activity has been well
established by Fabre,7 Domenjoz,8 Wilhelmi,9 and
Yourish.10 Lieberman11 reported on the effective
use of phenylbutazone in the treatment of painful
conditions of the musculoskeletal system in dogs;
including posterior paralysis associated with
intervertebral disc syndrome, painful fractures,
arthritis, and painful injuries to the limbs and joints.
Joshua12 observed objective improvement without
toxicity following long-term therapy of two aged
arthritic dogs. Ogilvie and Sutter13 reported rapid
response to phenylbutazone therapy in a review of
19 clinical cases including posterior paralysis,
posterior weakness, arthritis, rheumatism, and
other conditions associated with lameness and
musculoskeletal weakness.
Camberos14 reported favorable results withphenylbutazone following intermittent treatment of
Thoroughbred horses for arthritis and chronic
arthrosis (e.g., osteoarthritis of medial and distal
bones of the hock, arthritis of stifle and hip,
arthrosis of the spine, chronic hip pains, chronic
pain in trapezius muscles, and generalized
arthritis). Results were less favorable in cases of
traumatism, muscle rupture, strains and
inflammations of the third phalanx. Sutter15
reported favorable response in chronic equine
arthritis, fair results in a severely bruised mare, and
poor results in two cases where the condition was
limited to the third phalanx. - INDICATIONS
- CONTRAINDICATIONS
-
PRECAUTIONS
Stop medication at the first sign of
gastrointestinal upset, jaundice, or blood dyscrasia.
Authenticated cases of agranulocytosis associated
with the drug have occurred in man. To guard
against this possibility, conduct routine blood
counts at weekly intervals during the early phase of
therapy and at intervals of two weeks thereafter.
Any significant fall in the total white count, relative
decrease in granulocytes, or black or tarry stools,
should be regarded as a signal for immediate
cessation of therapy and institution of appropriate
counter measures. In the treatment of
inflammatory conditions associated with infections,
specific anti-infective therapy is required - STORAGE AND HANDLING
-
DOSAGE AND ADMINISTRATION
HORSES
INTRAVENOUSLY: 1 to 2 g per 1,000 lbs of body weight (5 to 10 mL/1,000 lbs) daily. Injection should be given slowly and with care. Limit intravenous administration to a maximum of 5 successive days, which may be followed by oral phenylbutazone dosage forms.GUIDELINES TO SUCCESSFUL THERAPY
1. Use a relatively high dose for the first 48 hours, then reduce gradually to a maintenance dose. Maintain lowest dose capable of producing desired clinical response.
2. Response to phenylbutazone therapy is prompt, usually occurring within 24 hours. If no significant clinical response is evident after 5 days, reevaluate diagnosis and therapeutic approach.
3. In animals, phenylbutazone is largely metabolized in 8 hours. It is recommended that a third of the daily dose be administered at 8 hour intervals. Reduce dosage as symptoms regress. In some cases, treatment may be given only when symptoms appear with no need for continuous medication. If long-term therapy is planned, oral administration is suggested.
4. Many chronic conditions will respond to phenylbutazone therapy, but discontinuance of treatment may result in recurrence of symptoms. -
CONTACT INFORMATION
To report suspected adverse events for technical assistance or to obtain a copy of the Safety Data Sheet, contact Sparhawk Laboratories Inc. at 1-800-255-6388 or 1-913-888-7500. For additional information about adverse drug experience reporting for animal drugs, contact FDA at 1-888-FDA-VETS or http://www.fda.gov/reportanimalae
- HOW SUPPLIED
-
REFERENCES
1. Kuzell, W.C., Schaffarzick, R.W., Naugler, W.G., and Mankle, E.A.: AMA Arch. Int. Med. 92:646, 1953.
2. Kuzell, W.C., Schaffarzick, R.W., Brown, B. and Mankle, E.A.: Jour. Amer. Med. Assoc. 149:729, 1952.
3. Kuzell, W.C., Schaffarzick, R.W., Calif. Med. 777:319, 1952.
4. Payne, R.W., Shetlar, M.R., Farr, C., Hellbaum, A.A., and Ishmael, W.K.T.: J.Lab. Clin. Med. 45:331, 1955.
5. Fleming, J. and @Will, G.: Ann Rheumat. Dis. 12:95, 1953.
6. Denko, C.W., and Rumi, D.: Amer. Practit. 6:1865, 1955.
7. Fabre, J. and Berger, A.: Semaine Hop. (Paris) 31:87, 1955.
8. Domenjoz, R., Theobald, W. and Morsdorf, K., Arzneimittel-Forsch. 5:488, 1955.
9 Wilhelmi, G., and Pulver, R.: Arzneimittel-Forsch. 5:221, 1955.
10. Yoursh, N., Paton, B., Brodie, B.B and Burns, J.J.: AMA Arch. Ophth. 53:264, 1955.
11. Lieberman, L.L.: Jour. Amer. Vet. Med. Assoc. 125:128, 1954.
12. Joshua, J.O.: Vet. Rec. 68:60 (Jan 21), 1956.
13. Ogilvie, F.B. and Sutter, M.D.: Vet. Med 52:492-494, 1957.
14. Camberos, H.R.: Rev. Med. Vet. (Buenos Aires); 38:9, 1956.
15. Sutter, M.F.: Vet Med. 53:83 (Feb.), 1958. -
INDICATIONS
For relief of inflammatory conditions associated with the musculoskeletal system in horses.
Intravenous dosage (not subcutaneous or intramuscular use):
Horses: 1 to 2 g per 1,000 lb body weight (5 to 10 mL/1,000 lb) daily.
Not for use in Horses intended for food.
See package outsert for additional information.
Approved by FDA under ANADA 200-371
Each mL contains:Phenylbutazone ............................. 200 mg
Benzyl Alcohol Preservative .........10.45 mg
Sodium hydroxide to adjust pH to 9.5 to 10.0
Water For Injection ...............................q.s. - PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EQUI-PHAR
phenylbutazone injectionProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:50989-425 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLBUTAZONE (UNII: GN5P7K3T8S) (PHENYLBUTAZONE - UNII:GN5P7K3T8S) PHENYLBUTAZONE 200 mg in 1 mL Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50989-425-12 100 mL in 1 VIAL Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANADA ANADA200371 07/01/2006 Labeler - Vedco, Inc. (021634266)




