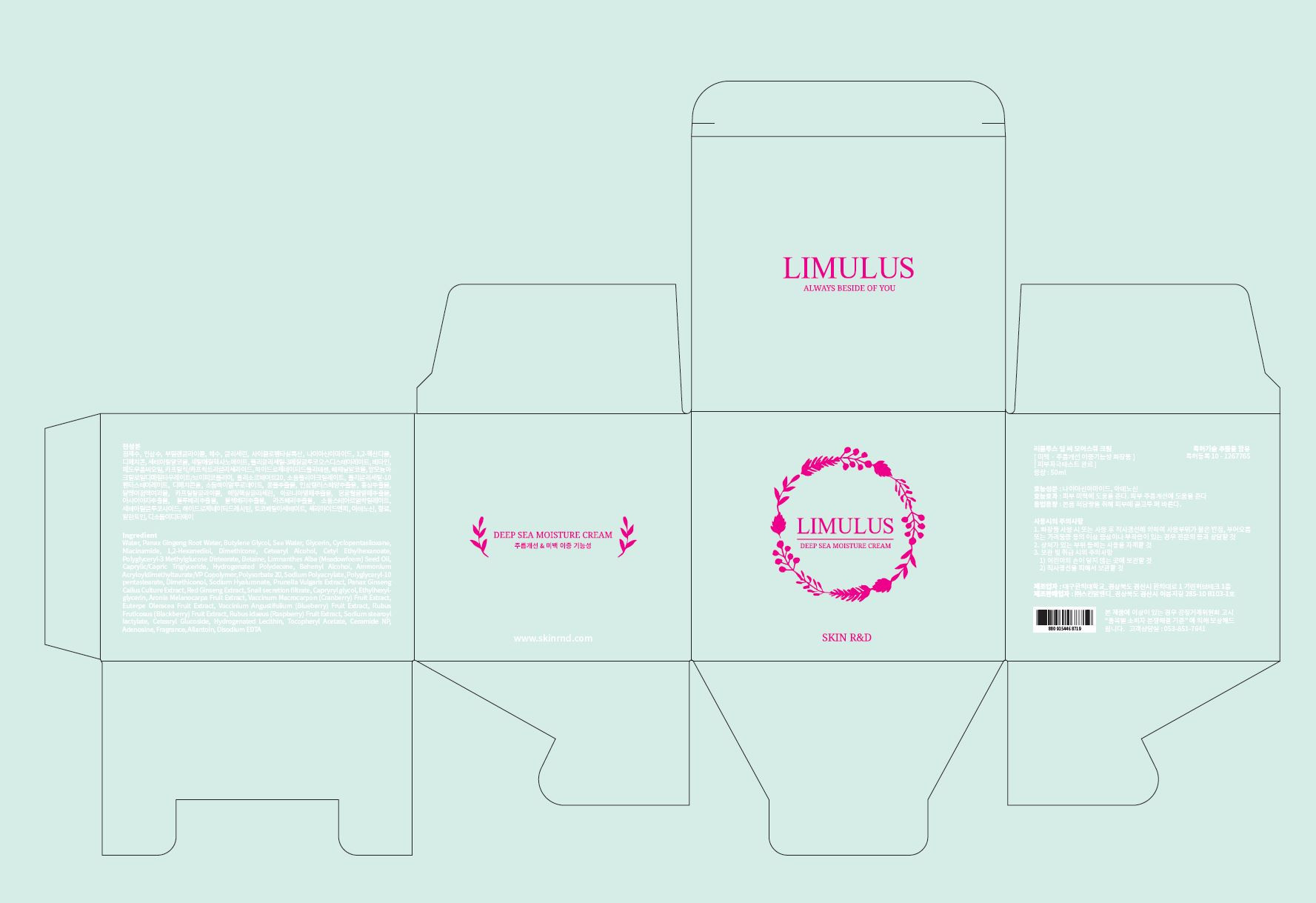
Label: LIMULUS DEEP SEA MOISTURE- niacinamide, adenosine cream
-
Contains inactivated NDC Code(s)
NDC Code(s): 71464-0001-1 - Packager: Skin R&D
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated June 29, 2017
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
-
INACTIVE INGREDIENT
Water
Panax Ginseng Root Water
Glycerin
1,2-Hexanediol
Butylene Glycol
Sea Water
Butylene Glycol
Glycerin
Cyclopentasiloxane
Dimethicone
1,2-Hexanediol
Cetearyl Alcohol
Cetyl Ethylhexanoate
Polyglyceryl-3 Methylglucose Distearate
Behenyl Alcohol
Polyglyceryl-10 pentastearate
Sodium Stearoyl Lactylate
Betaine
Limnanthes Alba (Meadowfoam) Seed Oil
Caprylic/Capric Triglyceride
Hydrogenated Polydecene
Cyclopentasiloxane
Dimethiconol
Cetearyl Alcohol
Cetearyl Glucoside
Panax Ginseng Callus Extract
Water
Glycerin
Butylene Glycol
1,2-Hexanediol
Aronia Melanocarpa Fruit Extract
Glycerin
Butylene Glycol
Snail secretion filtrate
Butylene Glycol
Water
Butylene Glycol
Vaccinium Macrocarpon (Cranberry) Fruit Extract
Euterpe Oleracea Fruit Extract
Vaccinium Angustifolium (Blueberry) Fruit Extract
Rubus Fruticosus (Blackberry) Fruit Extract
Rubus Idaeus (Raspberry) Fruit Extract
Prunella Vulgaris Extract
Butylene Glycol
Ammonium Acryloyldimethyltaurate/VP Copolymer
Polysorbate 20
Sodium Polyacrylate
Fragrance
Allantoin
Caprylyl glycol
Ethylhexylglycerin
Hydrogenated Lecithin
Tocopheryl Acetate
Ceramide NP
Disodium EDTA
Red Ginseng Extract
Sodium Hyaluronate - PURPOSE
- KEEP OUT OF REACH OF CHILDREN
- INDICATIONS & USAGE
-
WARNINGS
Precautions for Use
1. Consult with a medical specialist if any abnormal symptoms or side effects, such as red spots, swelling, or itching, occur during the use of this product.
2. Do not use the product on wounded areas.
3. Precautions for storage and handling
1) Keep out of reach of children.
2) Store away from direct sunlight.
- DOSAGE & ADMINISTRATION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LIMULUS DEEP SEA MOISTURE
niacinamide, adenosine creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:71464-0001 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ADENOSINE (UNII: K72T3FS567) (ADENOSINE - UNII:K72T3FS567) ADENOSINE 0.04 g in 100 mL NIACINAMIDE (UNII: 25X51I8RD4) (NIACINAMIDE - UNII:25X51I8RD4) NIACINAMIDE 2 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:71464-0001-1 50 mL in 1 JAR; Type 0: Not a Combination Product 05/01/2017 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 05/01/2017 Labeler - Skin R&D (694792365) Registrant - Skin R&D (694792365) Establishment Name Address ID/FEI Business Operations Daegu Haany University 690282954 manufacture(71464-0001) Establishment Name Address ID/FEI Business Operations Skin R&D 694792365 label(71464-0001)