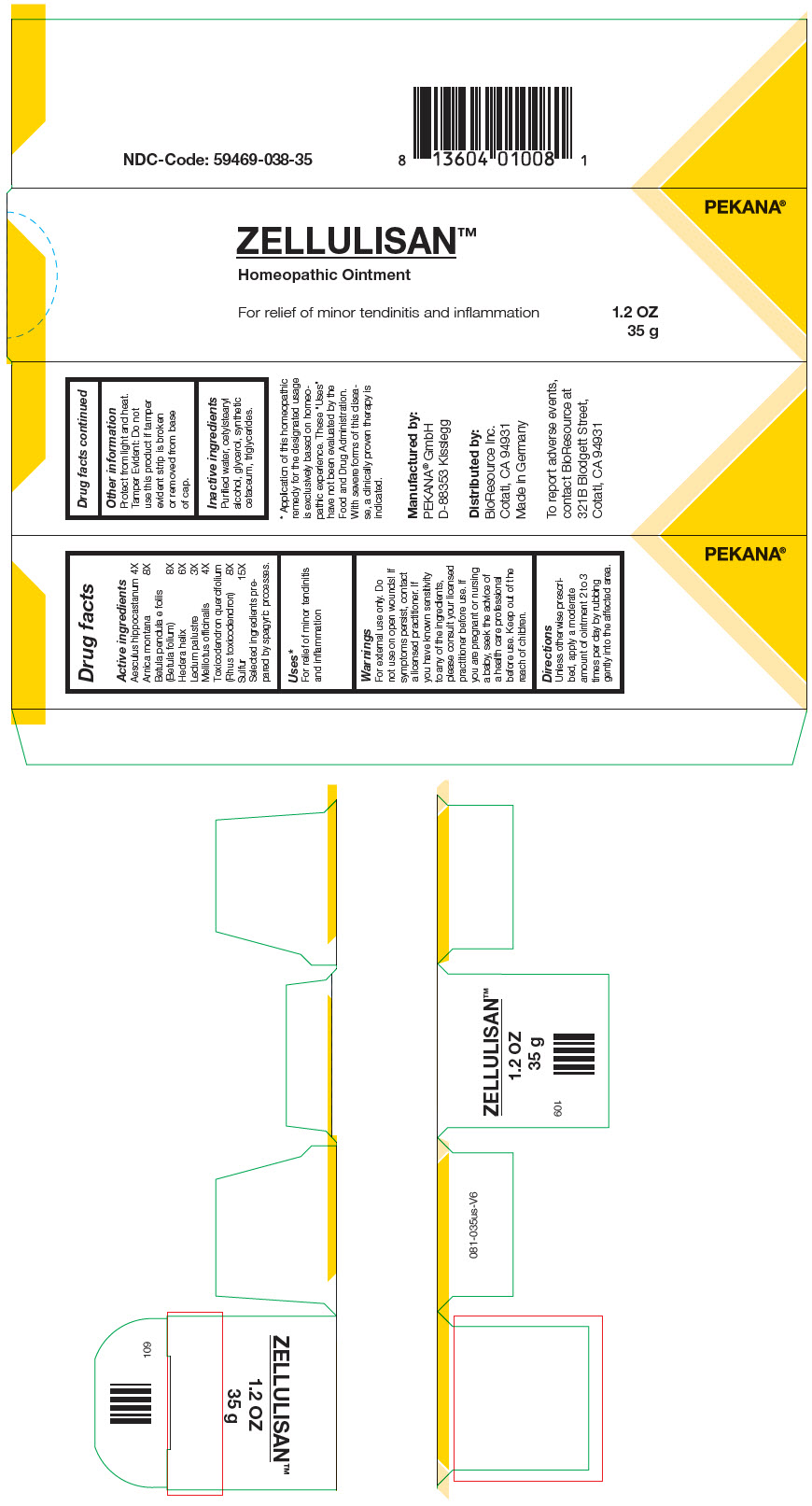
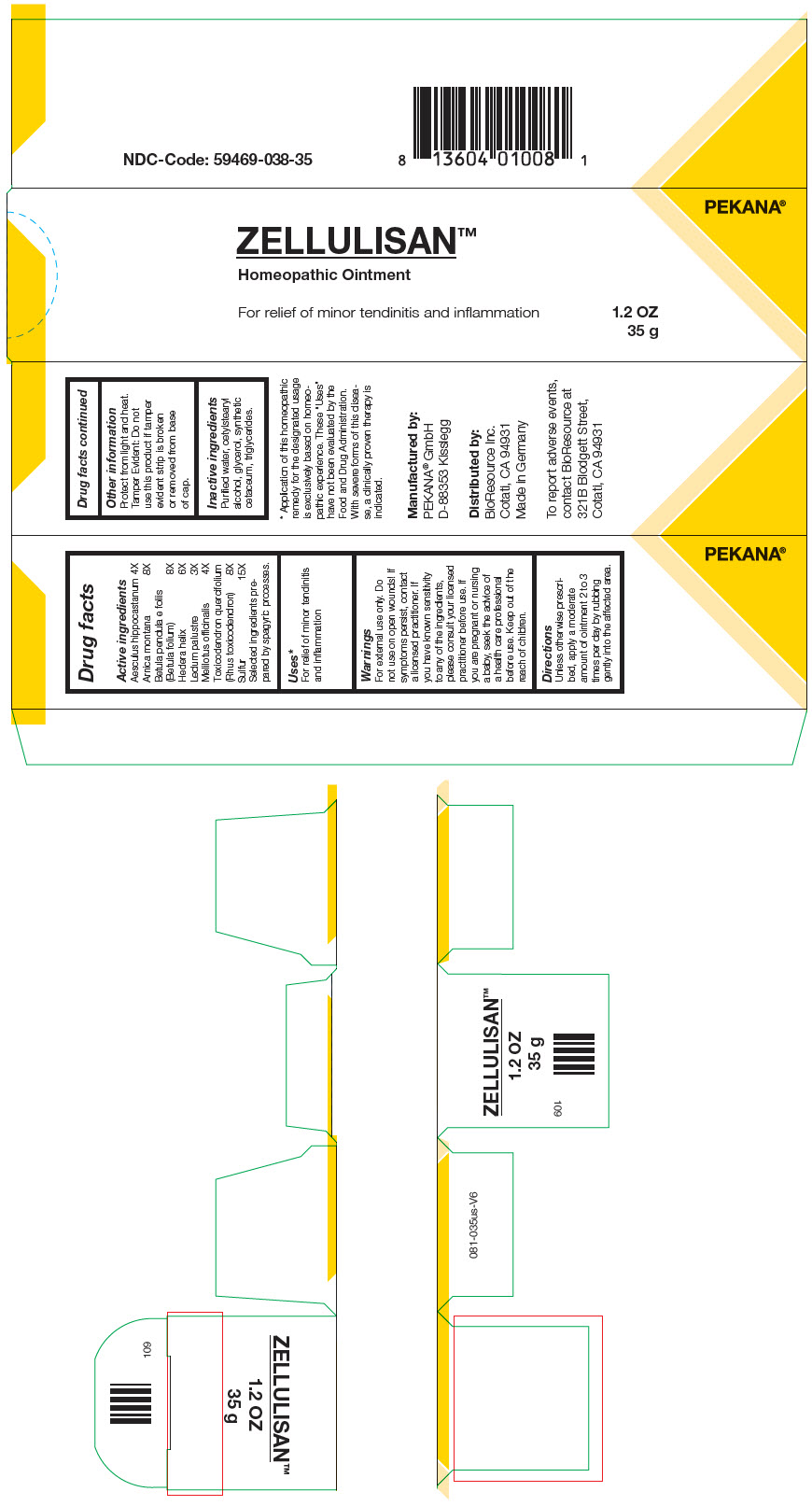
Label: ZELLULISAN- horse chestnut, arnica montana, betula pendula leaf, hedera helix flowering twig, ledum palustre twig, melilotus officinalis top, toxicodendron pubescens leaf, and sulfur ointment
- NDC Code(s): 59469-038-35
- Packager: Pekana-Naturheilmittel GmbH
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated December 21, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
-
Uses1
- 1
- Application of this homeopathic remedy for the designated usage is exclusively based on homeopathic experience. These "Uses" have not been evaluated by the Food and Drug Administration. With severe forms of this disease, a clinically proven therapy is indicated.
-
Warnings
For external use only. Do not use on open wounds! If symptoms persist, contact a licensed practitioner. If you have known sensitivity to any of the ingredients, please consult your licensed practitioner before use. If you are pregnant or nursing a baby, seek the advice of a health care professional before use.
- Directions
- Other information
- Inactive ingredients
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 35 g Tube Carton
-
INGREDIENTS AND APPEARANCE
ZELLULISAN
horse chestnut, arnica montana, betula pendula leaf, hedera helix flowering twig, ledum palustre twig, melilotus officinalis top, toxicodendron pubescens leaf, and sulfur ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:59469-038 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength horse chestnut (UNII: 3C18L6RJAZ) (horse chestnut - UNII:3C18L6RJAZ) horse chestnut 4 [hp_X] in 35 g ARNICA MONTANA WHOLE (UNII: O80TY208ZW) (ARNICA MONTANA WHOLE - UNII:O80TY208ZW) ARNICA MONTANA WHOLE 8 [hp_X] in 35 g betula pendula leaf (UNII: 5HW39H9KDH) (betula pendula leaf - UNII:5HW39H9KDH) betula pendula leaf 8 [hp_X] in 35 g hedera helix flowering twig (UNII: 3D10KUA6BM) (hedera helix flowering twig - UNII:3D10KUA6BM) hedera helix flowering twig 6 [hp_X] in 35 g RHODODENDRON TOMENTOSUM LEAFY TWIG (UNII: 877L01IZ0P) (RHODODENDRON TOMENTOSUM LEAFY TWIG - UNII:877L01IZ0P) RHODODENDRON TOMENTOSUM LEAFY TWIG 3 [hp_X] in 35 g melilotus officinalis top (UNII: GM6P02J2DX) (melilotus officinalis top - UNII:GM6P02J2DX) melilotus officinalis top 4 [hp_X] in 35 g toxicodendron pubescens leaf (UNII: 6IO182RP7A) (toxicodendron pubescens leaf - UNII:6IO182RP7A) toxicodendron pubescens leaf 8 [hp_X] in 35 g sulfur (UNII: 70FD1KFU70) (sulfur - UNII:70FD1KFU70) sulfur 15 [hp_X] in 35 g Inactive Ingredients Ingredient Name Strength water (UNII: 059QF0KO0R) cetostearyl alcohol (UNII: 2DMT128M1S) glycerin (UNII: PDC6A3C0OX) medium-chain triglycerides (UNII: C9H2L21V7U) cetyl esters wax (UNII: D072FFP9GU) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59469-038-35 1 in 1 CARTON 02/20/2011 1 35 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved homeopathic 02/20/2011 Labeler - Pekana-Naturheilmittel GmbH (320344542) Establishment Name Address ID/FEI Business Operations Pekana-Naturheilmittel GmbH 320344542 Manufacture(59469-038)