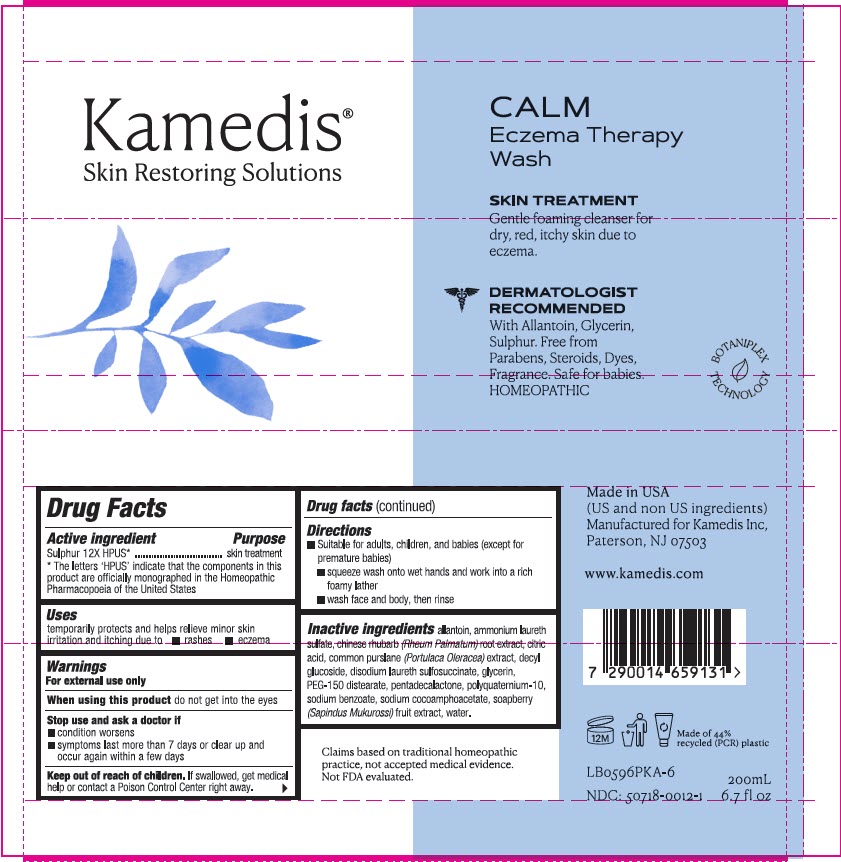
Label: CALM ECZEMA THERAPY WASH- sulphur 12x liquid
- NDC Code(s): 50718-0012-1
- Packager: Kamedis
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved homeopathic
DISCLAIMER: This homeopathic product has not been evaluated by the Food and Drug Administration for safety or efficacy. FDA is not aware of scientific evidence to support homeopathy as effective.
Drug Label Information
Updated January 30, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
Drug Facts
Active ingredient
Sulphur 12X HPUS*
* The letters 'HPUS' indicate that the components in this product are officially monographed in the Homeopathic Pharmacopoeia of the United StatesStop use and ask a doctor if
- condition worsens
- symptoms last more than 7 days or clear up and occur again within a few days
Keep out of reach of children.
- If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- Suitable for adults, children, and babies (except for premature babies)
- squeeze wash onto wet hands and work into a rich foamy lather
- wash face and body, then rinse
Inactive Ingredients
allantoin, ammonium laureth sulfate, Chinese rhubarb (Rheum palmatum) root extract, citric acid, common purslane (Portulaca oleracea) extract, decyl glucoside, disodium laureth sulfosuccinate, glycerin, PEG-150 distearate, pentadecalactone, polyquaternium-10, sodium benzoate, sodium cocoamphoacetate, soapberry (Sapindus mukurossi) fruit extract, water
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 200 mL Tube Label
-
INGREDIENTS AND APPEARANCE
CALM ECZEMA THERAPY WASH
sulphur 12x liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50718-0012 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SULFUR (UNII: 70FD1KFU70) (SULFUR - UNII:70FD1KFU70) SULFUR 12 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength ALLANTOIN (UNII: 344S277G0Z) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) DECYL GLUCOSIDE (UNII: Z17H97EA6Y) DISODIUM LAURETH SULFOSUCCINATE (UNII: D6DH1DTN7E) GLYCERIN (UNII: PDC6A3C0OX) PEG-150 DISTEARATE (UNII: 6F36Q0I0AC) PENTADECALACTONE (UNII: OK17S3S98K) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM COCOAMPHOACETATE (UNII: W7Q5E87674) WATER (UNII: 059QF0KO0R) RHEUM PALMATUM ROOT (UNII: G025DAL7CE) SAPINDUS MUKOROSSI FRUIT (UNII: 66H9NW427Y) PURSLANE (UNII: M6S840WXG5) AMMONIUM LAURETH-2 SULFATE (UNII: 698O4Z48G6) POLYQUATERNIUM-10 (10000 MPA.S AT 2%) (UNII: PI1STR9QYH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50718-0012-1 200 mL in 1 TUBE; Type 0: Not a Combination Product 01/01/2018 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 01/01/2018 Labeler - Kamedis (080311300) Establishment Name Address ID/FEI Business Operations Biogenesis Inc. 069117328 manufacture(50718-0012)