Label: SANITIZING WIPES- benzalkonium chloride swab
- NDC Code(s): 72202-200-01, 72202-200-24
- Packager: Zogics, LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated November 29, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredients
-
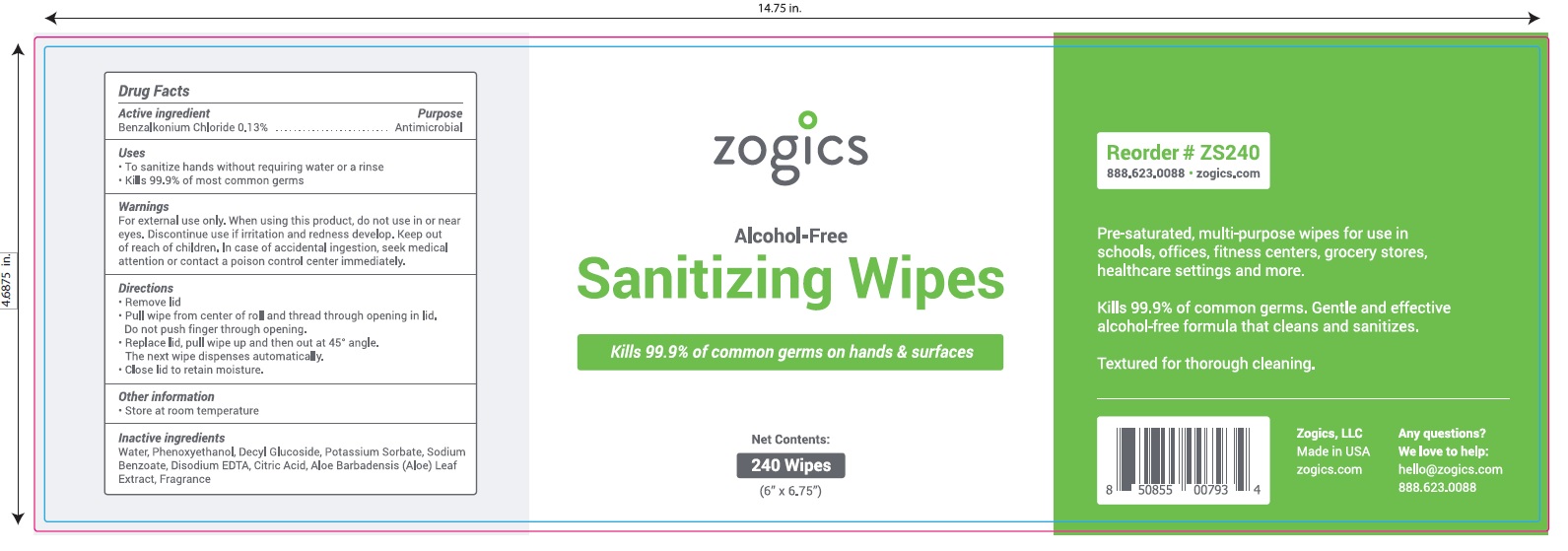
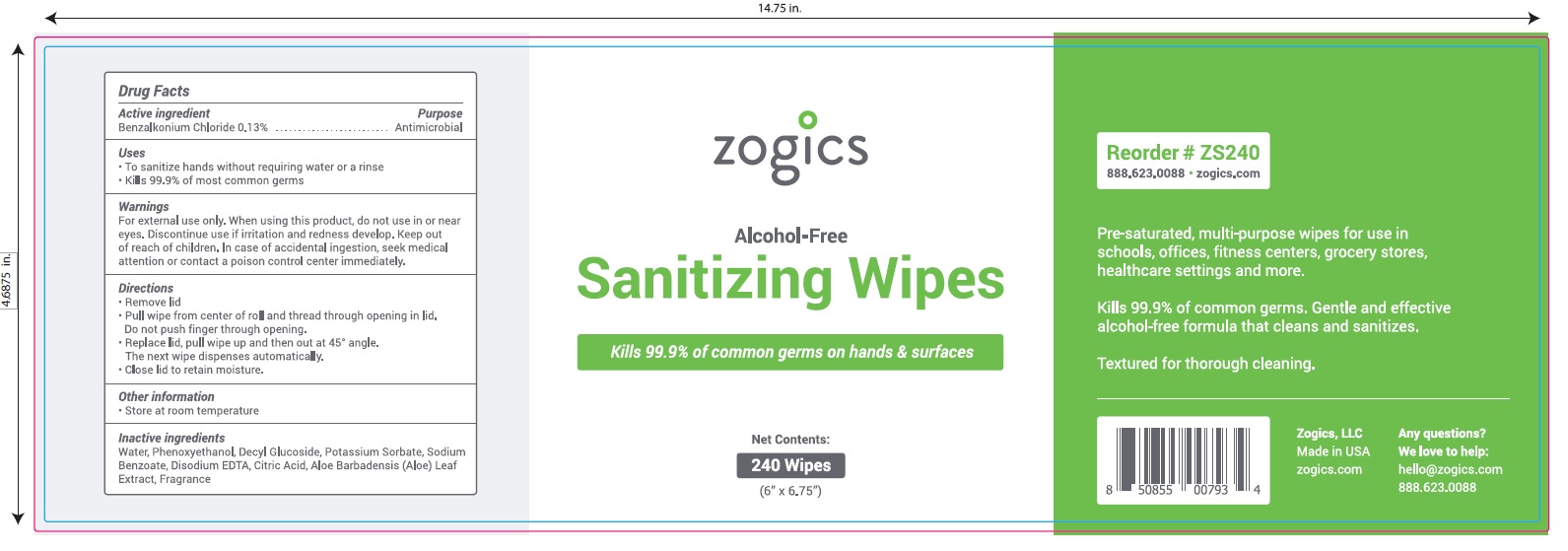
Principal Display Panel
NDC 72202-200-01
2000 count
zogics Sanitizing Wipes
Reorder# Z2000 888.623.0088
Pre-saturated, multipurpose wipes for use in school, offices, fitness centers, grocery stores, healthcare settings and more.
Kills 99.9% of most common germs.
Gentle and effective alcohol-free formula that cleans and sanitizes.
Testured for thorough cleaning
Net Content: 2000 Wipes per Roll (8"X5.25")
Zogics, LLC Made in USA
Any Questions? We love to help: hello@zogicz.com or 888.623.0088

NDC 72202-200-24
zogics
Alcohol- Free
Sanitizing Wipes
Kills 99.9% of common germs on hands and surfaces.
Pre-saturated, multi-purpose wipes for use in schools, offices, fitness centers, grocery stores, healthcare settings and more.
Kills 99.9% of common germs.
Gentle and effective alcohol-free formula that cleans and sanitizes
Textured for thorough cleaning.
Net Contents: 240 Wipes (6" x 6.75")
Zogics, LLC Made in USA
Any Questions? We love to help: hello@zogicz.com or 888.623.0088
-
INGREDIENTS AND APPEARANCE
SANITIZING WIPES
benzalkonium chloride swabProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:72202-200 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.13 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PHENOXYETHANOL (UNII: HIE492ZZ3T) DECYL GLUCOSIDE (UNII: Z17H97EA6Y) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SODIUM BENZOATE (UNII: OJ245FE5EU) EDETATE DISODIUM (UNII: 7FLD91C86K) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) ALOE VERA LEAF (UNII: ZY81Z83H0X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:72202-200-01 2000 in 1 BAG 04/02/2018 1 1 g in 1 PACKET; Type 0: Not a Combination Product 2 NDC:72202-200-24 240 in 1 BAG 04/02/2018 2 1 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 04/02/2018 Labeler - Zogics, LLC (796321870) Registrant - Zogics, LLC (796321870) Establishment Name Address ID/FEI Business Operations Precare Corp. 117111327 manufacture(72202-200)