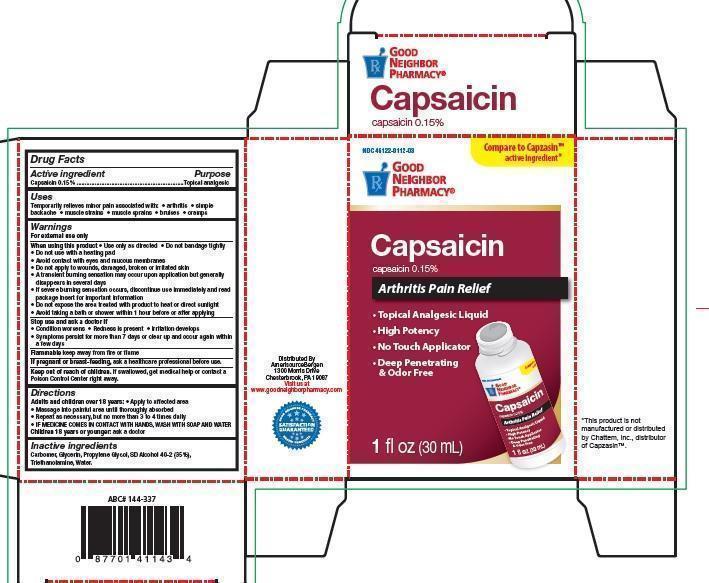
Label: GOOD NEIGHBOR PHARMACY CAPSAICIN- capsaicin liquid
-
Contains inactivated NDC Code(s)
NDC Code(s): 46122-112-03 - Packager: Amerisourcebergen Drug Corporation
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 22, 2015
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Purpose
- Uses
-
Warnings
For external use only
When Using this product
• Use only as directed • Do not bandage tightly
• Do not use with a heating pad
• Avoid contact with eyes and mucous membranes
• Do not apply to wounds, damaged, broken or irritated skin
• A transient burning sensation may occur upon application but generally
disappears in several days
• If severe burning sensation occurs, discontinue use immediately and read
package insert for important information
• Do not expose the area treated with product to heat or direct sunlight
• Avoid taking a bath or shower within 1 hour before or after applying
- Stop use and ask a doctor if
- Flammable
- If pregnant or breast-feeding,
- Keep out of reach of children
- Directions
- Inactive ingredients
- SPL UNCLASSIFIED SECTION
- Good Neighbor Pharmacy Capsaicin
-
INGREDIENTS AND APPEARANCE
GOOD NEIGHBOR PHARMACY CAPSAICIN
capsaicin liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:46122-112 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CAPSAICIN (UNII: S07O44R1ZM) (CAPSAICIN - UNII:S07O44R1ZM) CAPSAICIN 150 mg in 100000 mg Inactive Ingredients Ingredient Name Strength CARBOMER 940 (UNII: 4Q93RCW27E) Glycerin (UNII: PDC6A3C0OX) Propylene Glycol (UNII: 6DC9Q167V3) ALCOHOL (UNII: 3K9958V90M) TROLAMINE (UNII: 9O3K93S3TK) Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:46122-112-03 150 mg in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part348 10/28/2011 Labeler - Amerisourcebergen Drug Corporation (007914906) Establishment Name Address ID/FEI Business Operations Product Quest MFG, LLC 927768135 manufacture(46122-112) , label(46122-112)