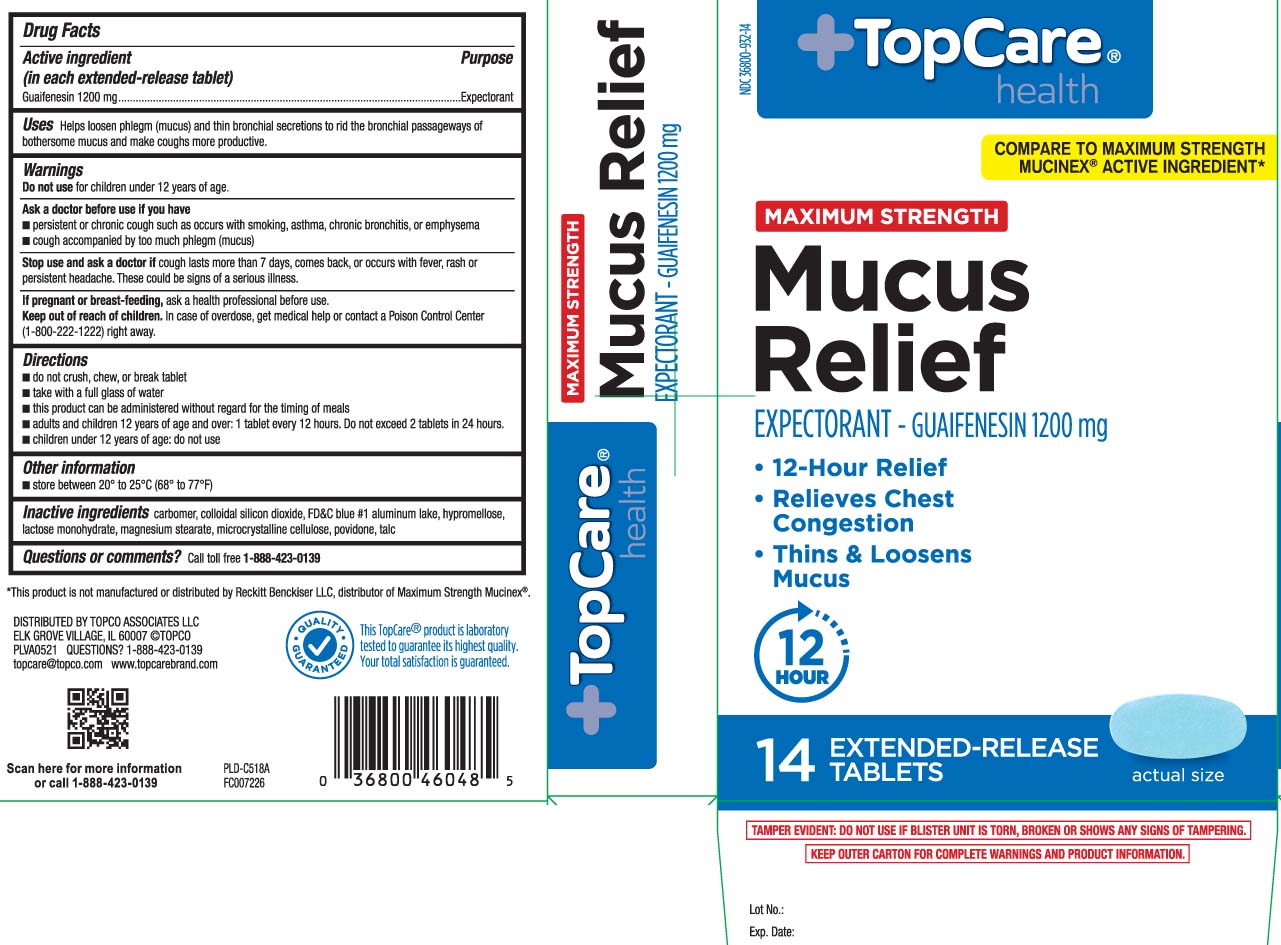
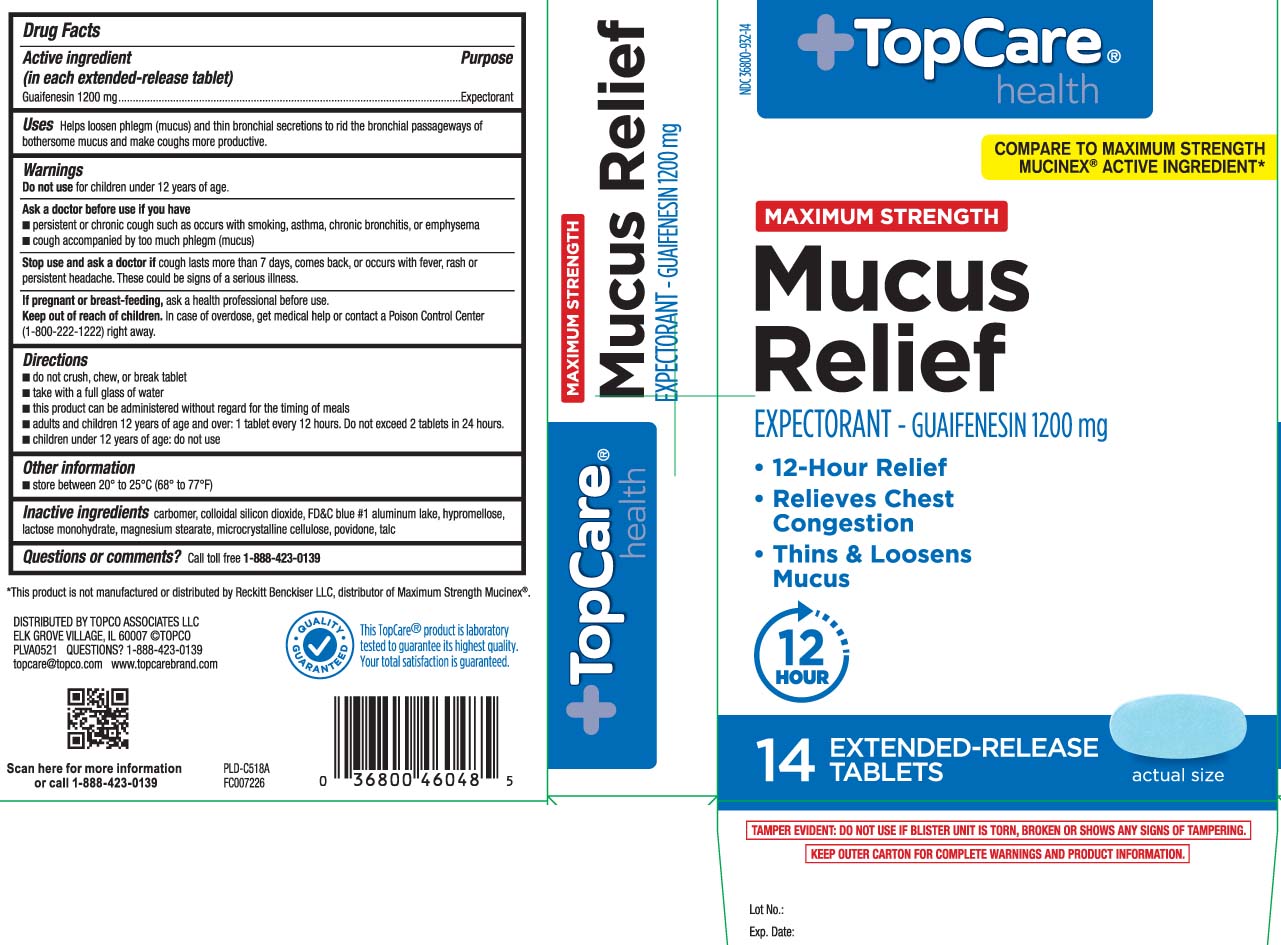
Label: MUCUS RELIEF MAXIMUM STRENGTH- guaifenesin tablet
- NDC Code(s): 36800-932-14
- Packager: TOP CARE (Topco Associates LLC)
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated November 28, 2022
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each extended-release tablet)
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- persistent or chronic cough such as occurs with smoking, asthma, chronic bronchitis or emphysema
- cough accopanied by too much phlegm (mucus)
- Directions
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
COMPARE TO MAXIMUM STRENGTH MUCINEX® ACTIVE INGREDIENT*
MAXIMUM STRENGTH
Mucus Relief
EXPECTORANT - GUAIFENESIN 1200 mg
- 12-Hour Relief
- Relieves Chest Congestion
- Thins & Loosens Mucus
EXTENDED-RELEASE TABLETS
*This product is not manufactured or distributed by Reckitt Benckiser LLC, distributor of Maximum Strength Mucinex®.
TAMPER EVIDENT: DO NOT USE IF BLISTER UNIT IS TORN, BROKEN OR SHOWS ANY SIGNS OF TAMPERING.
KEEP OUTER CARTON FOR COMPLETE WARNINGS AND PRODUCT INFORMATION.
DISTRIBUTED BY TOPCO ASSOCIATES LLC,
ELK GROVE VILLAGE, IL 60007
- Package Label
-
INGREDIENTS AND APPEARANCE
MUCUS RELIEF MAXIMUM STRENGTH
guaifenesin tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:36800-932 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength GUAIFENESIN (UNII: 495W7451VQ) (GUAIFENESIN - UNII:495W7451VQ) GUAIFENESIN 1200 mg Inactive Ingredients Ingredient Name Strength SILICON DIOXIDE (UNII: ETJ7Z6XBU4) MAGNESIUM STEARATE (UNII: 70097M6I30) CARBOMER 934 (UNII: Z135WT9208) CELLULOSE, MICROCRYSTALLINE (UNII: OP1R32D61U) POVIDONE (UNII: FZ989GH94E) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) TALC (UNII: 7SEV7J4R1U) Product Characteristics Color blue Score no score Shape OVAL Size 22mm Flavor Imprint Code AN037 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:36800-932-14 14 in 1 CARTON 03/31/2019 1 1 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA207342 03/31/2019 Labeler - TOP CARE (Topco Associates LLC) (006935977)