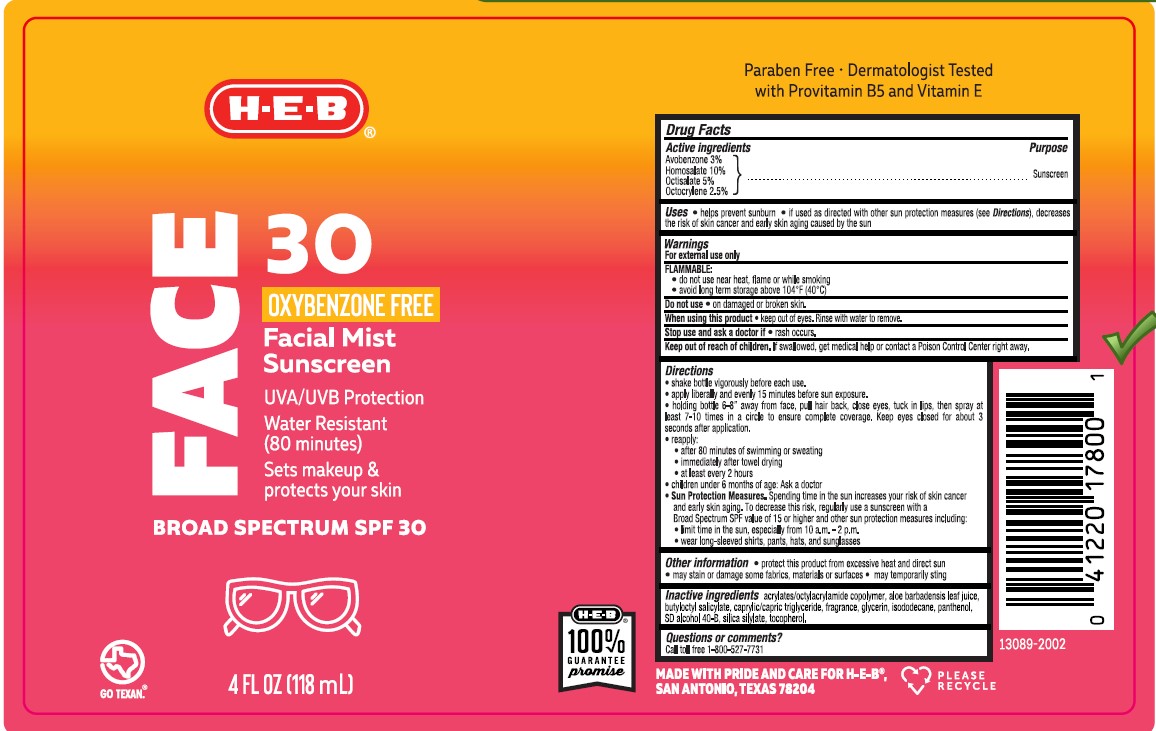
Label: HEB SOLUTIONS SUNSCREEN FACE 50 FACIAL MIST SUNSCREEN- avobenzone, homosalate, octisalate, octocrylene spray
- NDC Code(s): 37808-127-04
- Packager: H.E.B
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 15, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredients
- Purpose
- Uses
- Warnings
- FLAMMABLE:
- Do not use
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children.
-
Directions
- shake bottle vigorously before each use.
- apply liberally and evenly 15 minutes before sun exposure.
- hold bottle 6-8 inches away from face, pull hair back, close eyes, tuck lips, then spray at least 7-10 times in a circle to ensure complete coverage. Keep eyes closed for about 3 seconds after application.
- reapply:
• after 80 minutes of swimming or sweating
• immediately after towel drying
• at least every 2 hours
- children under 6 months of age: Ask a doctor
- Sun Protection Measures. Spending time in the sun increases your risk of skin cancer and early skin aging. To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
• limit time in the sun, especially from 10 a.m. - 2 p.m.
• wear long-sleeved shirts, pants, hats, and sunglasses
- Other information
- Inactive ingredients
- Label
-
INGREDIENTS AND APPEARANCE
HEB SOLUTIONS SUNSCREEN FACE 50 FACIAL MIST SUNSCREEN
avobenzone, homosalate, octisalate, octocrylene sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:37808-127 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength AVOBENZONE (UNII: G63QQF2NOX) (AVOBENZONE - UNII:G63QQF2NOX) AVOBENZONE 30 mg in 1 mL HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 100 mg in 1 mL OCTISALATE (UNII: 4X49Y0596W) (OCTISALATE - UNII:4X49Y0596W) OCTISALATE 50 mg in 1 mL OCTOCRYLENE (UNII: 5A68WGF6WM) (OCTOCRYLENE - UNII:5A68WGF6WM) OCTOCRYLENE 25 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALCOHOL (UNII: 3K9958V90M) ACRYLATE/ISOBUTYL METHACRYLATE/N-TERT-OCTYLACRYLAMIDE COPOLYMER (75000 MW) (UNII: JU3XHR8VWK) ALOE VERA LEAF (UNII: ZY81Z83H0X) .ALPHA.-TOCOPHEROL, DL- (UNII: 7QWA1RIO01) GLYCERIN (UNII: PDC6A3C0OX) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) BUTYLOCTYL SALICYLATE (UNII: 2EH13UN8D3) ISODODECANE (UNII: A8289P68Y2) PANTHENOL (UNII: WV9CM0O67Z) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:37808-127-04 118 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product 03/02/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 03/02/2020 Labeler - H.E.B (007924756)