Label: CREST PRO-HEALTH SENSITIVE AND GUM ALL DAY PROTECTION- stannous fluoride paste, dentifrice
- NDC Code(s): 69423-765-16, 69423-765-37
- Packager: The Procter & Gamble Manufacturing Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 12, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purposes
-
Uses
- aids in the prevention of cavities
- helps prevent gingivitis
- helps interfere with the harmful effects of plaque associated with gingivitis
- helps control plaque bacteria that contribute to the development of gingivitis
- builds increasing protection against painful sensitivity of the teeth to cold, heat, acids, sweets or contact
- Warnings
-
Directions
- adults and children 12 yrs. & older: apply at least a 1-inch strip of the product onto a soft bristle toothbrush. Brush teeth thoroughly for at least 1 minute twice a day (morning and evening) or as recommended by a dentist. Make sure to brush all sensitive areas of the teeth.
- do not swallow
- children under 12 yrs.: ask a dentist
-
Other information
- products containing stannous fluoride may produce surface staining of the teeth
- adequate toothbrushing may prevent these stains which are not harmful or permanent and may be removed by your dentist
- this Crest is specially formulated to help prevent staining
- see your dentist regularly
Apply full brush head of paste. First brush sensitive areas for 30 seconds and finish brushing the rest of the mouth thoroughly.
- Inactive ingredients
- Questions?
- SPL UNCLASSIFIED SECTION
-
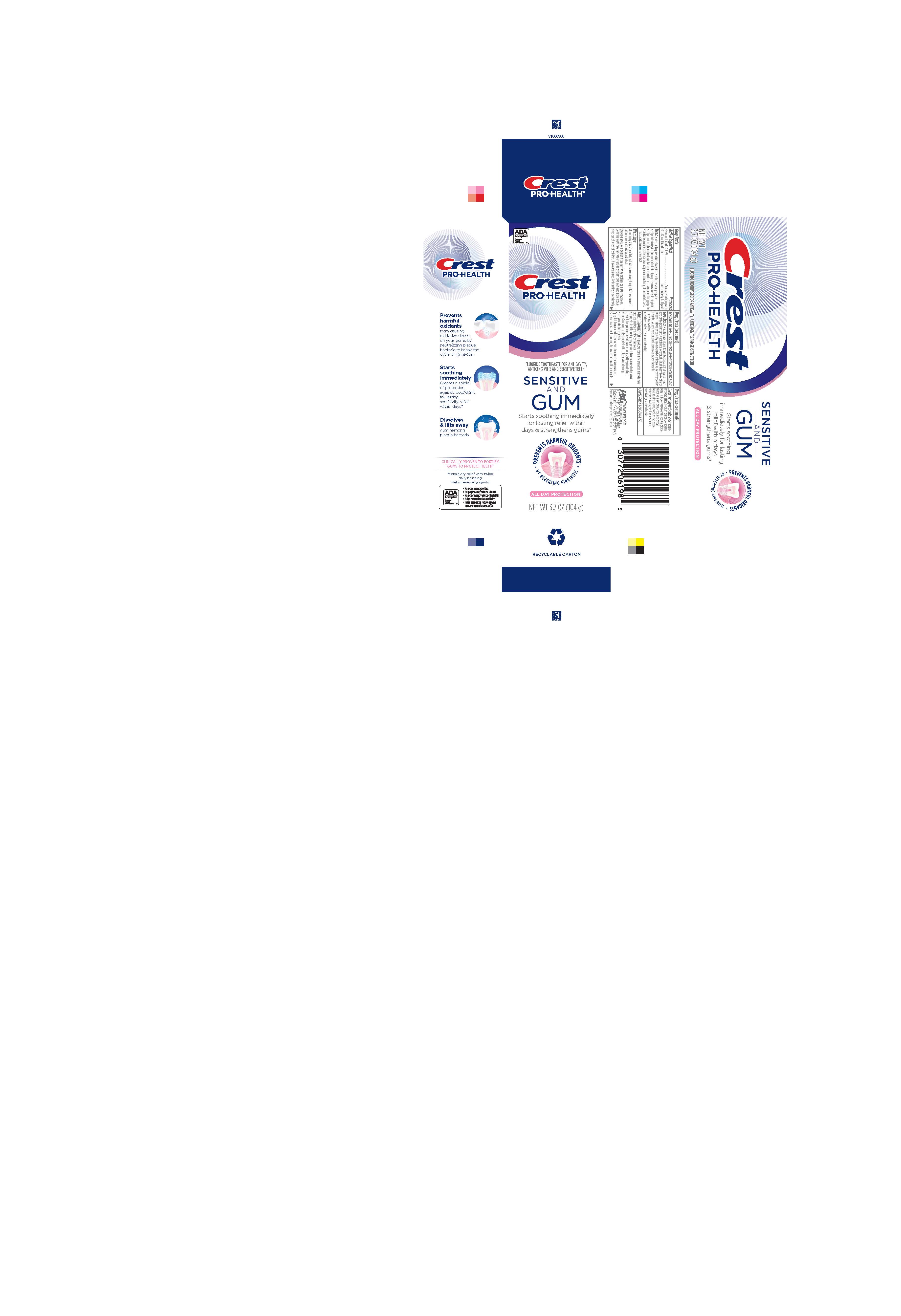
PRINCIPAL DISPLAY PANEL - 104 g Tube Carton
ADA Accepted
American Dental Association
Crest
PRO-HEALTH
FLUORIDE TOOTHPASTE FOR ANTICAVITY, ANTIGINGIVITIS AND SENSITIVE TEETH
SENSITIVE AND GUM
Starts soothing immediately for lasting relief within days & strengthens gums*
PREVENTS HARMFUL OXIDANTS
BY REVERSING GINGIVITIS
ALL DAY PROTECTION*
NET WT 3.7 OZ (104 g)
-
INGREDIENTS AND APPEARANCE
CREST PRO-HEALTH SENSITIVE AND GUM ALL DAY PROTECTION
stannous fluoride paste, dentifriceProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69423-765 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength STANNOUS FLUORIDE (UNII: 3FTR44B32Q) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 1.5 mg in 1 g Inactive Ingredients Ingredient Name Strength SUCRALOSE (UNII: 96K6UQ3ZD4) HYDRATED SILICA (UNII: Y6O7T4G8P9) SORBITOL (UNII: 506T60A25R) SODIUM CITRATE (UNII: 1Q73Q2JULR) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) WATER (UNII: 059QF0KO0R) SODIUM LAURYL SULFATE (UNII: 368GB5141J) SODIUM GLUCONATE (UNII: R6Q3791S76) CARRAGEENAN (UNII: 5C69YCD2YJ) SACCHARIN SODIUM (UNII: SB8ZUX40TY) XANTHAN GUM (UNII: TTV12P4NEE) ZINC CITRATE (UNII: K72I3DEX9B) SODIUM HYDROXIDE (UNII: 55X04QC32I) STANNOUS CHLORIDE (UNII: 1BQV3749L5) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white Score Shape Size Flavor MINT Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69423-765-37 1 in 1 CARTON 02/16/2023 1 104 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:69423-765-16 1 in 1 CELLO PACK 02/16/2023 2 1 in 1 CARTON 2 104 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M021 02/16/2023 Labeler - The Procter & Gamble Manufacturing Company (004238200)

