Label: NICOTINE TRANSDERMAL SYSTEM STEP 1- nicotine patch, extended release
NICOTINE TRANSDERMAL SYSTEM STEP 2- nicotine patch, extended release
-
Contains inactivated NDC Code(s)
NDC Code(s): 63868-190-14, 63868-191-14 - Packager: Chain Drug Marketing Association Inc.
- Category: HUMAN OTC DRUG LABEL
Drug Label Information
Updated June 19, 2014
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each patch)
- Purpose
- Use
-
Warnings
If you are pregnant or breast-feeding, only use this medicine on the advice of your health care provider. Smoking can seriously harm your child. Try to stop smoking without using any nicotine replacement medicine. This medicine is believed to be safer than smoking. However, the risks to your child from this medicine are not fully known.
- Do not use
- Ask a doctor before use if you have
- Ask a doctor or a pharmacist before use if you are
- When using this product
- Stop use and ask a doctor if
- Keep out of reach of children and pets.
-
Directions
- if you are under 18 years of age, ask a doctor before use
- before using this product, read the enclosed self-help guide for complete directions and other information
- stop smoking completely when you begin using the patch
- if you smoke more than 10 cigarettes per day, use the following schedule:
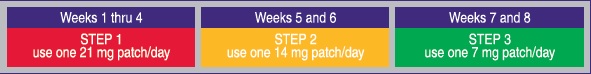
- if you smoke 10 or less cigarettes per day, start with Step 2 for 6 weeks, then Step 3 for 2 weeks and then stop
- apply one new patch every 24 hours on skin that is dry, clean and hairless
- remove backing from patch and immediately press onto skin. Hold for 10 seconds.
- wash hands after applying or removing patch. Save pouch to use for patch disposal. Dispose of used patch by folding sticky ends together and putting in pouch.
- the used patch should be removed and a new one applied to a different skin site at the same time each day
- if you have vivid dreams, you may remove the patch at bedtime and apply a new one in the morning
- do not wear more than one patch at a time
- do not cut patch in half or into smaller pieces
- do not leave patch on for more than 24 hours because it may irritate your skin and loses strength after 24 hours
- stop using the patch at the end of 8 weeks. If you still feel the need to use the patch talk to your doctor.
- Other information
- Inactive ingredients
- Comments or questions?
-
Principal Display Panel
NDC 63868-190-14
*Compare to the active ingredient in HABITROL®
Clear
Nicotine
Transdermal System
Step 1Stop Smoking Aid
Nicotine/21 mg Delivered Over 24 Hours
Includes:
Behavior Support Program with Self-Help Guide
Free Audio CD Upon Request. See self-help guide inside.If you Smoke MORE than 10 cigarrettes per day:
Start with Step 1
If you smoke 10 or LESS cigarrettes per day:
Start with Step 221 mg
14 Pactches©Distributed by
C.D.M.A, Inc.
Novi, MI 48376-0995
www.qualitychoice.com
Questions: 248-449-9300
-
Principal Display Panel
NDC 63868-190-14
*Compare to the active ingredient in HABITROL®
Clear
Nicotine
Transdermal System
Step 2Stop Smoking Aid
Nicotine/21 mg Delivered Over 24 Hours
Includes:
Behavior Support Program with Self-Help Guide
Free Audio CD Upon Request. See self-help guide inside.If you Smoke MORE than 10 cigarrettes per day:
Start with Step 1
If you smoke 10 or LESS cigarrettes per day:
Start with Step 214 mg
14 Pactches©Distributed by
C.D.M.A, Inc.
Novi, MI 48376-0995
www.qualitychoice.com
Questions: 248-449-9300
-
INGREDIENTS AND APPEARANCE
NICOTINE TRANSDERMAL SYSTEM STEP 1
nicotine patch, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63868-190 Route of Administration TRANSDERMAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NICOTINE (UNII: 6M3C89ZY6R) (NICOTINE - UNII:6M3C89ZY6R) NICOTINE 21 mg in 24 h Inactive Ingredients Ingredient Name Strength HEPTANE (UNII: 456148SDMJ) XYLENE (UNII: D856J1047R) ETHYL ACETATE (UNII: 76845O8NMZ) ISOPROPYL ALCOHOL (UNII: ND2M416302) TOLUENE (UNII: 3FPU23BG52) ETHYLBENZENE (UNII: L5I45M5G0O) VINYL ACETATE (UNII: L9MK238N77) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63868-190-14 14 in 1 CARTON 1 1 in 1 POUCH 1 24 h in 1 PATCH Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA074612 10/01/1999 NICOTINE TRANSDERMAL SYSTEM STEP 2
nicotine patch, extended releaseProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63868-191 Route of Administration TRANSDERMAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength NICOTINE (UNII: 6M3C89ZY6R) (NICOTINE - UNII:6M3C89ZY6R) NICOTINE 14 mg in 24 h Inactive Ingredients Ingredient Name Strength HEPTANE (UNII: 456148SDMJ) XYLENE (UNII: D856J1047R) ETHYL ACETATE (UNII: 76845O8NMZ) ISOPROPYL ALCOHOL (UNII: ND2M416302) TOLUENE (UNII: 3FPU23BG52) ETHYLBENZENE (UNII: L5I45M5G0O) VINYL ACETATE (UNII: L9MK238N77) HIGH DENSITY POLYETHYLENE (UNII: UG00KM4WR7) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63868-191-14 14 in 1 CARTON 1 1 in 1 POUCH 1 24 h in 1 PATCH Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA074612 10/01/1999 Labeler - Chain Drug Marketing Association Inc. (011920774)


