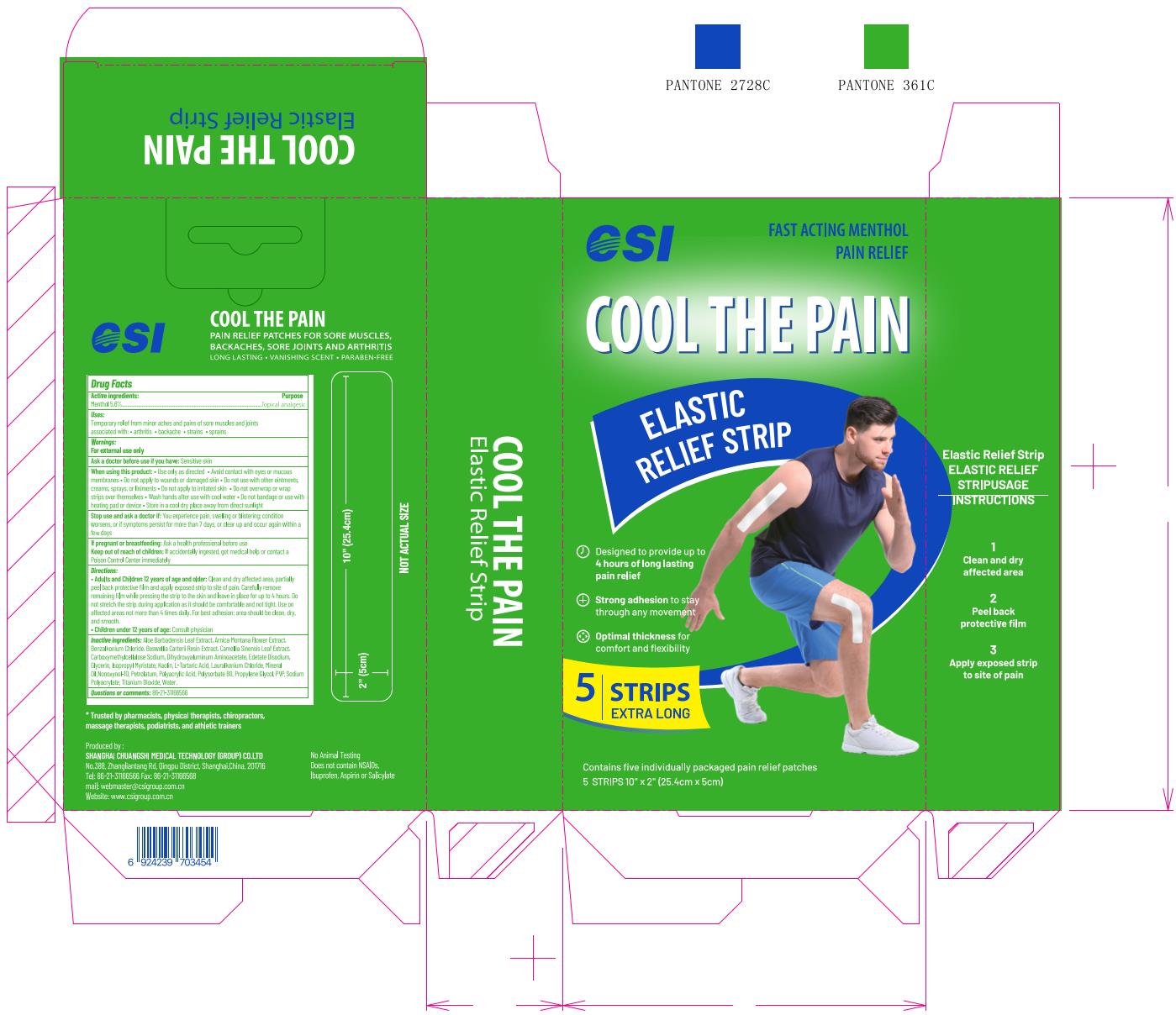
Label: ELASTIC RELIEF STRIP- menthol patch
- NDC Code(s): 73557-130-01, 73557-130-05
- Packager: Shanghai Chuangshi Medical Technology (Group) Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 29, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
- For external use only
- Use only as directed
- Avoid contact with eyes or mucous membranes
- Do not apply to wounds or damaged skin
- Do not use with other ointments, creams, sprays, or liniments
- Do not apply to irritated skin
- Do not overwrap or wrap strips over themselves
- Do not bandage or use with heating pad or device
- Ask doctor
-
When using
- Use only as directed
- Avoid contact with eyes or mucous membranes
- Do not apply to wounds or damaged skin
- Do not use with other ointments, creams, sprays, or liniments
- Do not apply to irritated skin
- Do not overwrap or wrap strips over themselves
- Wash hands after use with cool water
- Do not bandage or use with heating pad or device
- Do not use
- Stop use
- Pregnancy or breast feeding
- Keep out of reach of children
-
Directions
- Adult and Children 12 years of age and older: Clean and dry affected area, partially peel back protective film and apply exposed strip to site of pain. Carefully remove remaining film while pressing the strip to the skin and leave in place for up to 4 hours. Do not strech the strip during application as it should be comfortable and not tight. Use on affected areas not more than 4 times daily. For best adhesion: area should be clean, dry, and smooth.
- Children under 12 years of age: Consult physician
- Dosage forms & strengths
- Inactive ingredients
- Questions
- Other information
- Package label. Principal display panel
-
INGREDIENTS AND APPEARANCE
ELASTIC RELIEF STRIP
menthol patchProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73557-130 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 0.056 g in 1 g Inactive Ingredients Ingredient Name Strength POLYSORBATE 80 (UNII: 6OZP39ZG8H) 0.018 g in 1 g GLYCERIN (UNII: PDC6A3C0OX) 0.24 g in 1 g POVIDONE K90 (UNII: RDH86HJV5Z) 0.012 g in 1 g GREEN TEA LEAF (UNII: W2ZU1RY8B0) 0.0001 g in 1 g ALOE VERA LEAF (UNII: ZY81Z83H0X) 0.0001 g in 1 g EDETATE DISODIUM (UNII: 7FLD91C86K) 0.001 g in 1 g ARNICA MONTANA FLOWER (UNII: OZ0E5Y15PZ) 0.0001 g in 1 g FRANKINCENSE (UNII: R9XLF1R1WM) 0.0001 g in 1 g KAOLIN (UNII: 24H4NWX5CO) 0.001 g in 1 g TARTARIC ACID (UNII: W4888I119H) 0.001 g in 1 g WATER (UNII: 059QF0KO0R) 0.30808 g in 1 g DIHYDROXYALUMINUM AMINOACETATE ANHYDROUS (UNII: 1K713C615K) 0.0015 g in 1 g PROPYLENE GLYCOL (UNII: 6DC9Q167V3) 0.07 g in 1 g POLYACRYLIC ACID (250000 MW) (UNII: 9G2MAD7J6W) 0.2 g in 1 g CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) 0.001 g in 1 g SODIUM POLYACRYLATE (2500000 MW) (UNII: 05I15JNI2J) 0.05 g in 1 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) 0.001 g in 1 g MINERAL OIL (UNII: T5L8T28FGP) 0.03 g in 1 g PETROLATUM (UNII: 4T6H12BN9U) 0.002 g in 1 g BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) 0.001 g in 1 g LAURALKONIUM CHLORIDE (UNII: 07HUP5A29X) 0.001 g in 1 g NONOXYNOL-10 (UNII: K7O76887AP) 0.00002 g in 1 g ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) 0.005 g in 1 g Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73557-130-05 5 in 1 BOX 12/30/2022 1 NDC:73557-130-01 9 g in 1 PATCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 12/30/2022 Labeler - Shanghai Chuangshi Medical Technology (Group) Co., Ltd. (546872672) Registrant - Shanghai Chuangshi Medical Technology (Group) Co., Ltd. (546872672) Establishment Name Address ID/FEI Business Operations Shanghai Chuangshi Medical Technology (Group) Co., Ltd. 546872672 manufacture(73557-130) , label(73557-130)