Label: PAIN RELIEF GEL-ROLL ON- lidocaine and menthol gel
- NDC Code(s): 73557-373-25
- Packager: Shanghai Chuangshi Medical Technology (Group) Co., Ltd.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 29, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Use
- Warnings
- Do not use
- When using this product
- Stop use if
- Ask a doctor if
- Flammable
- If pregnant or breast-feeding
- Keep out of reach of children
- Directions
- Inactive Ingredients
- Questions or Comments
- GENERAL PRECAUTIONS
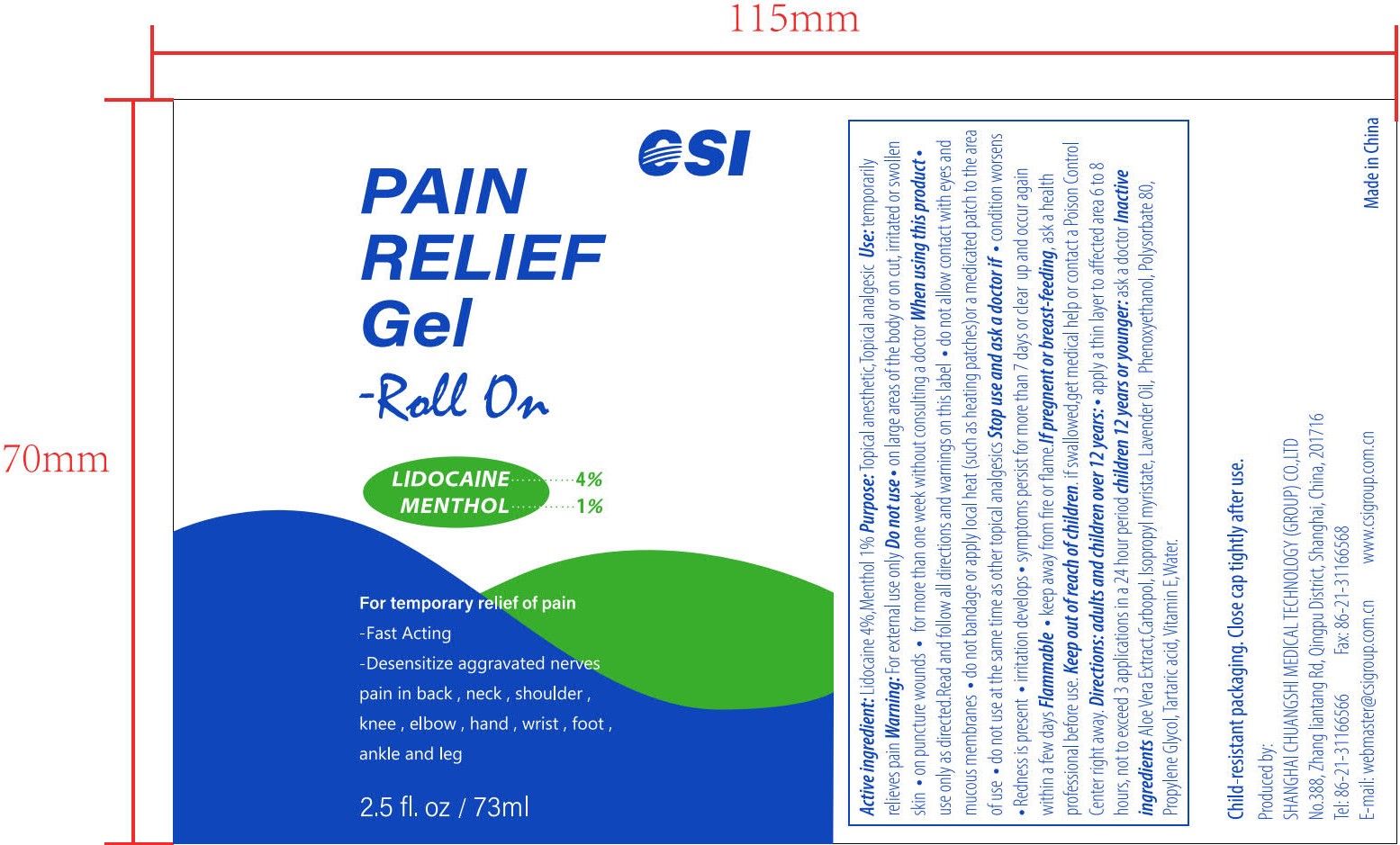
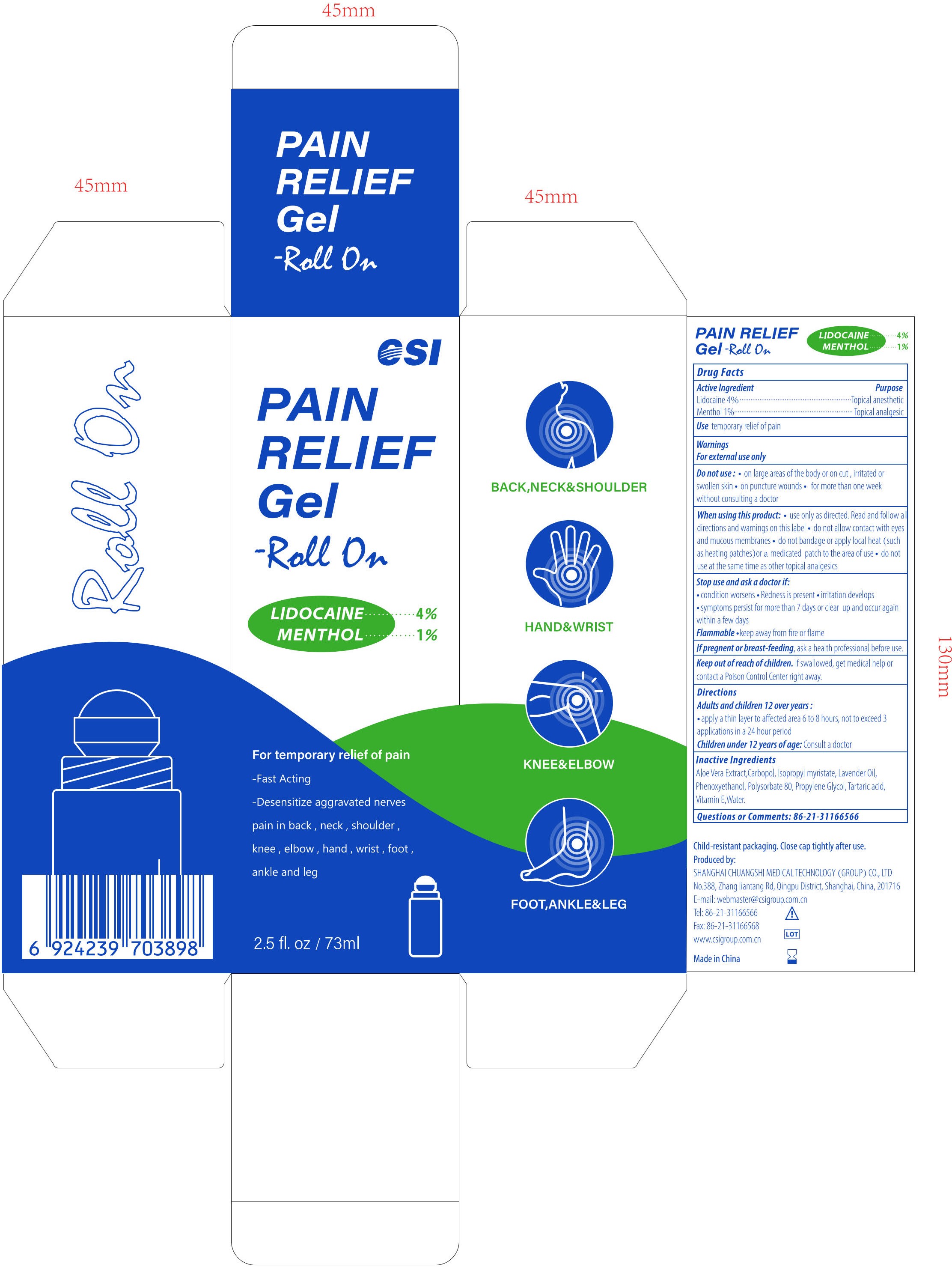
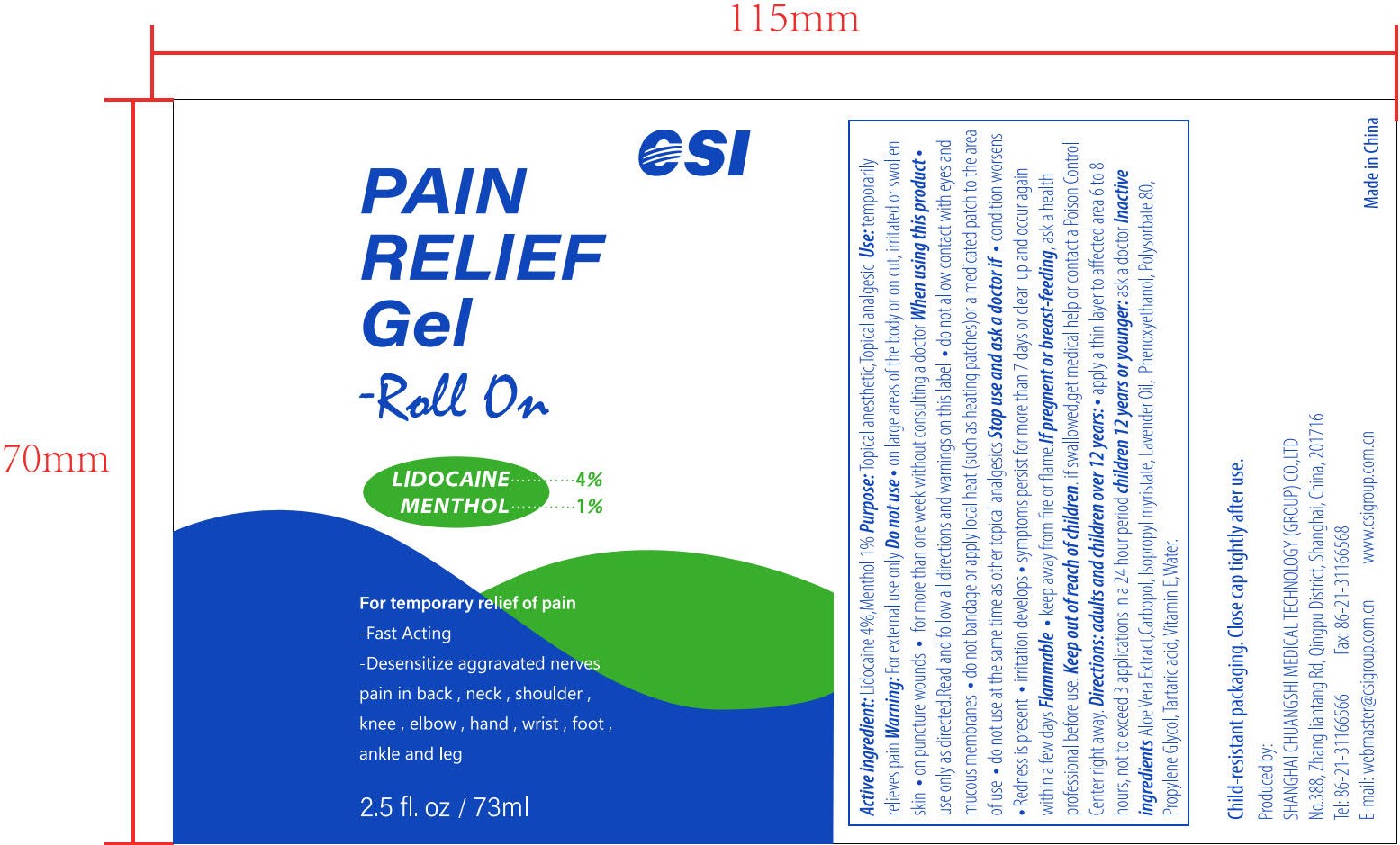
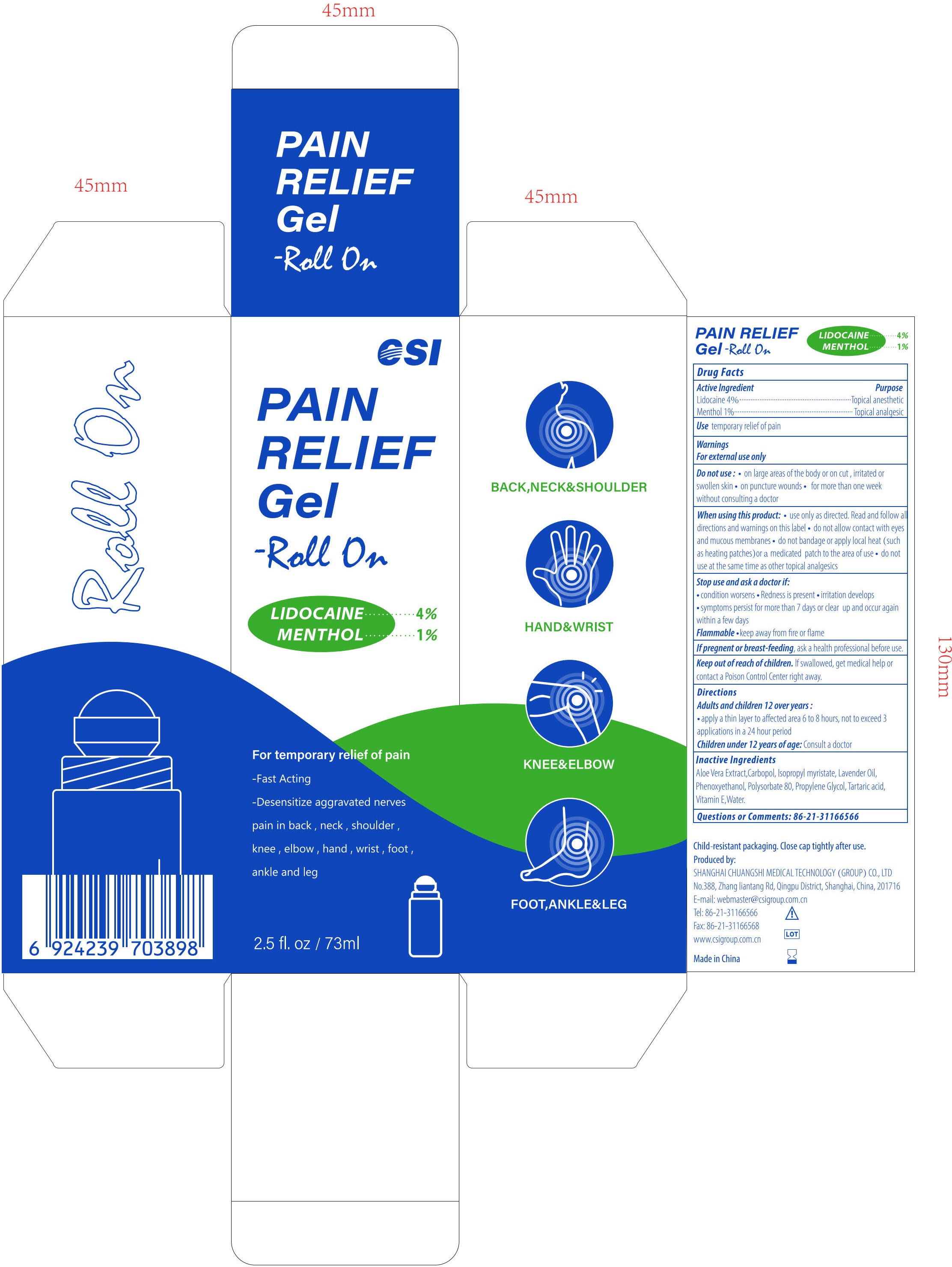
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
PAIN RELIEF GEL-ROLL ON
lidocaine and menthol gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:73557-373 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE (UNII: 98PI200987) (LIDOCAINE - UNII:98PI200987) LIDOCAINE 0.04 g in 1 mL MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 0.01 g in 1 mL Inactive Ingredients Ingredient Name Strength POLYACRYLIC ACID (250000 MW) (UNII: 9G2MAD7J6W) WATER (UNII: 059QF0KO0R) LAVENDER OIL (UNII: ZBP1YXW0H8) PHENOXYETHANOL (UNII: HIE492ZZ3T) ISOPROPYL MYRISTATE (UNII: 0RE8K4LNJS) .ALPHA.-TOCOPHEROL, D- (UNII: N9PR3490H9) ALOE VERA LEAF (UNII: ZY81Z83H0X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) TARTARIC ACID (UNII: W4888I119H) POLYSORBATE 80 (UNII: 6OZP39ZG8H) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:73557-373-25 73 mL in 1 BOTTLE; Type 0: Not a Combination Product 12/22/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 12/22/2022 Labeler - Shanghai Chuangshi Medical Technology (Group) Co., Ltd. (546872672) Registrant - Shanghai Chuangshi Medical Technology (Group) Co., Ltd. (546872672) Establishment Name Address ID/FEI Business Operations Shanghai Chuangshi Medical Technology (Group) Co., Ltd. 546872672 manufacture(73557-373) , label(73557-373)