Label: PAIN RELIEVER- acetaminophen tablet
- NDC Code(s): 68016-541-01
- Packager: Chain Drug Consortium
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated May 12, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
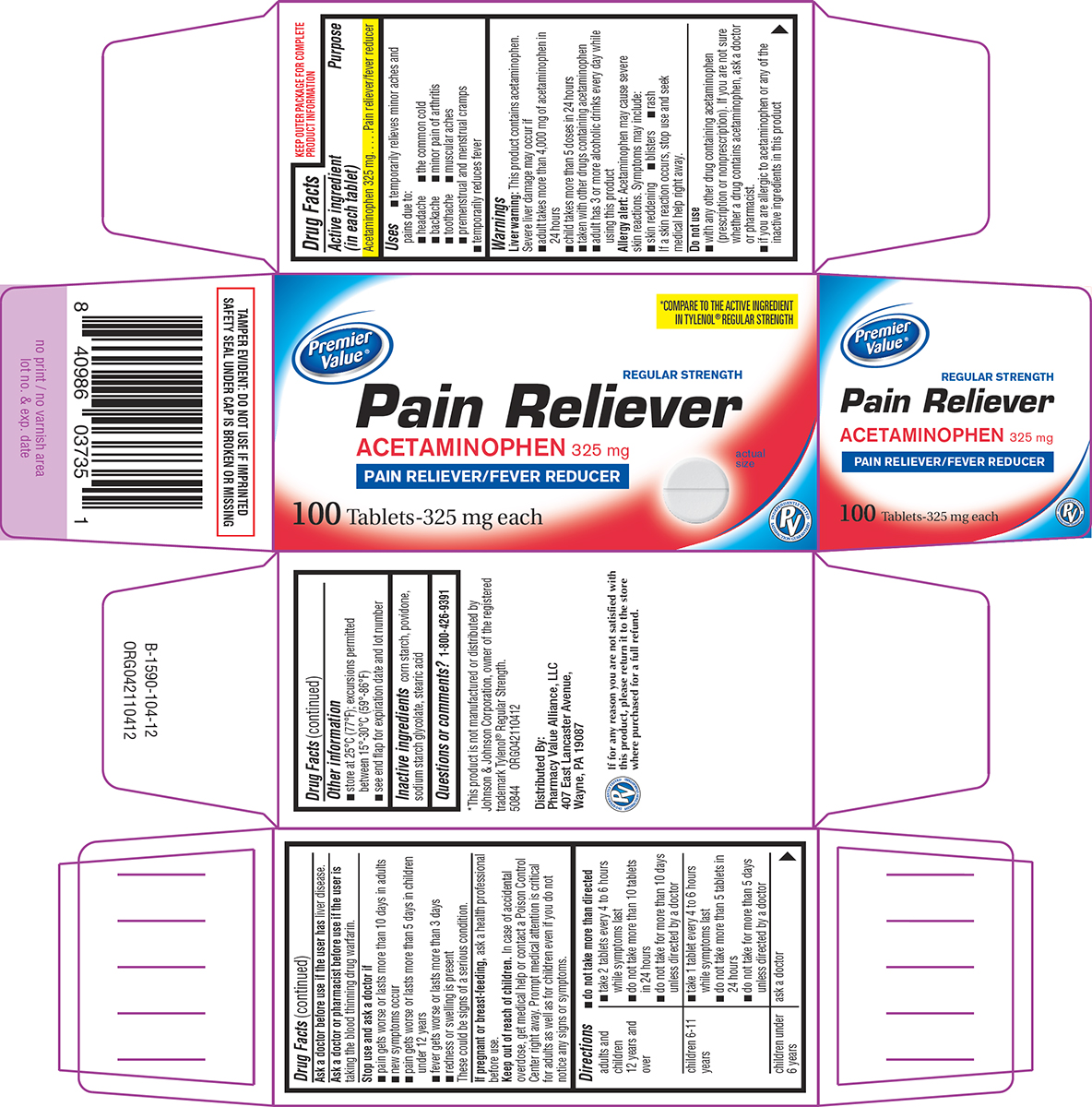
- Active ingredient (in each tablet)
- Purpose
- Uses
-
Warnings
Liver warning: This product contains acetaminophen. Severe liver damage may occur if
- adult takes more than 4,000 mg of acetaminophen in 24 hours
- child takes more than 5 doses in 24 hours
- taken with other drugs containing acetaminophen
- adult has 3 or more alcoholic drinks every day while using this product
Allergy alert: Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are allergic to acetaminophen or any of the inactive ingredients in this product
-
Directions
- do not take more than directed
adults and children 12 years and over ■ take 2 tablets every 4 to 6 hours while symptoms last
■ do not take more than 10 tablets in 24 hours
■ do not take for more than 10 days unless directed by a doctorchildren 6-11 years ■ take 1 tablet every 4 to 6 hours while symptoms last
■ do not take more than 5 tablets in 24 hours
■ do not take for more than 5 days unless directed by a doctorchildren under 6 years ask a doctor
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal display panel
Premier
Value®*COMPARE TO THE ACTIVE INGREDIENT
IN TYLENOL® REGULAR STRENGTHREGULAR STRENGTH
Pain Reliever
ACETAMINOPHEN 325 mg
PAIN RELIEVER/FEVER REDUCERactual
size100 Tablets-325 mg each
TAMPER EVIDENT: DO NOT USE IF IMPRINTED
SAFETY SEAL UNDER CAP IS BROKEN OR MISSING*This product is not manufactured or distributed by
Johnson & Johnson Corporation, owner of the registered
trademark Tylenol® Regular Strength.
50844 ORG042110412Distributed By:
Pharmacy Value Alliance, LLC
407 East Lancaster Avenue,
Wayne, PA 19087If for any reason you are not satisfied with
this product, please return it to the store
where purchased for a full refund.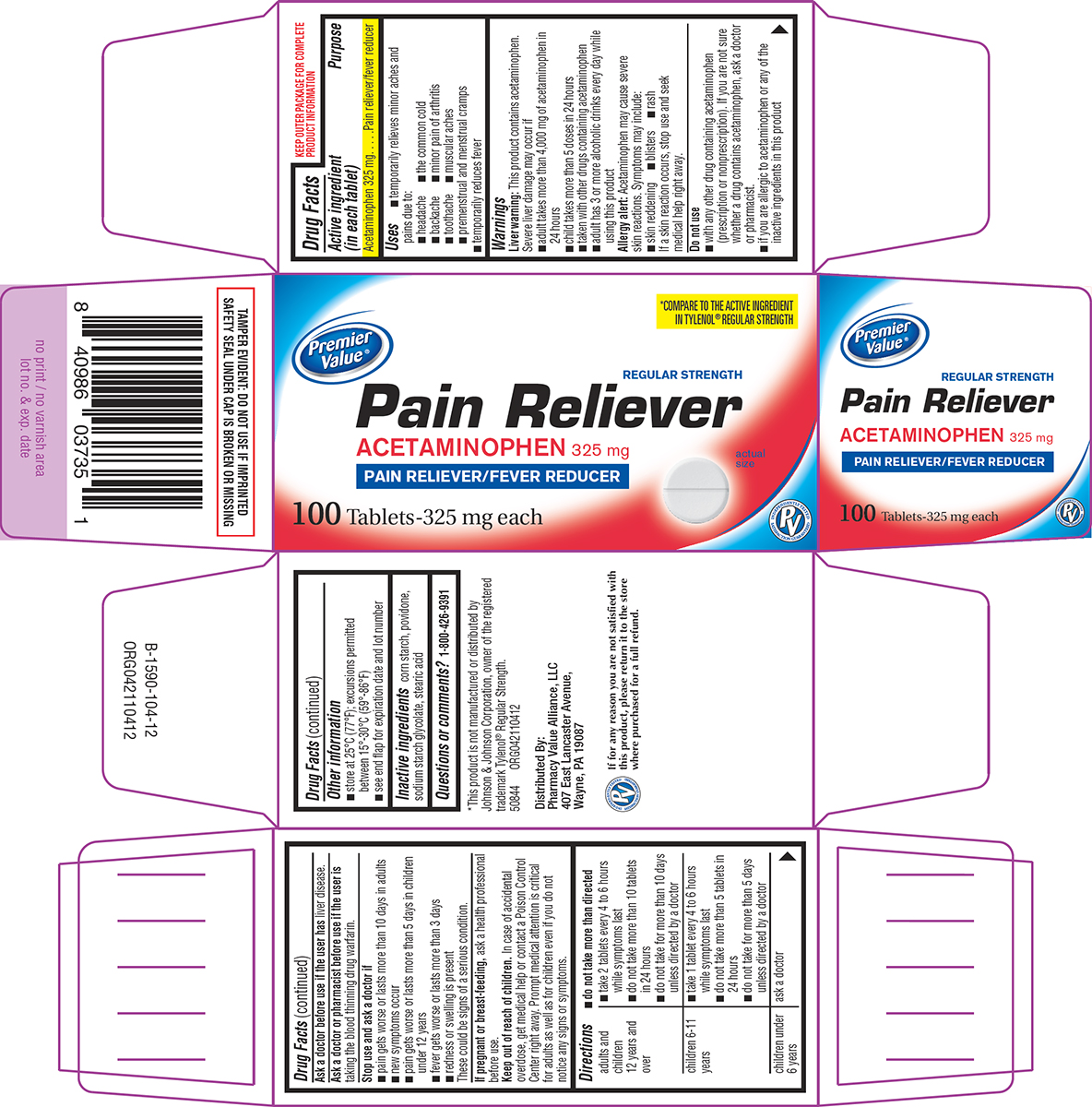
Premier Value 44-104
-
INGREDIENTS AND APPEARANCE
PAIN RELIEVER
acetaminophen tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68016-541 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) SODIUM STARCH GLYCOLATE TYPE A POTATO (UNII: 5856J3G2A2) STEARIC ACID (UNII: 4ELV7Z65AP) Product Characteristics Color white Score 2 pieces Shape ROUND Size 10mm Flavor Imprint Code 44;104 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68016-541-01 1 in 1 CARTON 05/12/2023 1 100 in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M013 05/12/2023 Labeler - Chain Drug Consortium (101668460) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 038154464 pack(68016-541) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 832867837 manufacture(68016-541) Establishment Name Address ID/FEI Business Operations LNK International, Inc. 967626305 pack(68016-541)