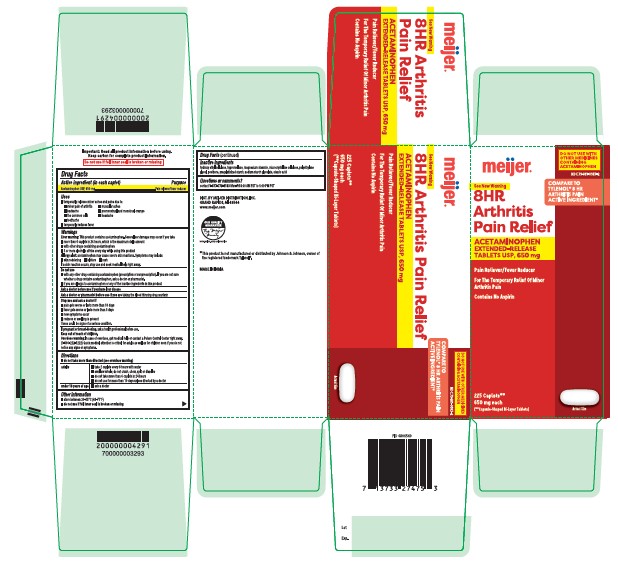
Label: ACETAMINOPHEN tablet
- NDC Code(s): 79481-0183-2, 79481-0183-4, 79481-0183-5
- Packager: MEIJER DISTRIBUTION INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated December 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient (in each caplet)
- Purpose
- Uses
- Liver Warning
- Allergy Alert
- Do not use
- Ask a doctor before use if you have
- Ask a doctor or pharmacist before use if you are
- Stop use and ask a doctor if
- If pregnant or breast-feeding,
- Keep out of reach of children.
- Overdose warning:
-
Directions
■ do not take more than directed (see overdose warning)
adults ■ take 2 caplets every 8 hours with water
■ swallow whole; do not crush, chew, split or dissolve
■ do not take more than 6 caplets in 24 hours
■ do not use for more than 10 days unless directed by a doctorunder 18 years of age ■ ask a doctor
- Other Information
- Inactive ingredients
- Questions or comments?
- PDP
-
INGREDIENTS AND APPEARANCE
ACETAMINOPHEN
acetaminophen tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79481-0183 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 650 mg Inactive Ingredients Ingredient Name Strength POVIDONE K30 (UNII: U725QWY32X) STARCH, CORN (UNII: O8232NY3SJ) SODIUM STARCH GLYCOLATE TYPE A CORN (UNII: AG9B65PV6B) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) MAGNESIUM STEARATE (UNII: 70097M6I30) HYDROXYETHYL CELLULOSE (140 CPS AT 5%) (UNII: 8136Y38GY5) STEARIC ACID (UNII: 4ELV7Z65AP) HYPROMELLOSE 2910 (6 MPA.S) (UNII: 0WZ8WG20P6) Product Characteristics Color white Score no score Shape CAPSULE Size 19mm Flavor Imprint Code G650 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79481-0183-5 150 in 1 BOTTLE; Type 0: Not a Combination Product 07/17/2023 2 NDC:79481-0183-2 225 in 1 BOTTLE; Type 0: Not a Combination Product 07/17/2023 3 NDC:79481-0183-4 400 in 1 BOTTLE; Type 0: Not a Combination Product 07/17/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA211544 07/17/2023 Labeler - MEIJER DISTRIBUTION INC (006959555)