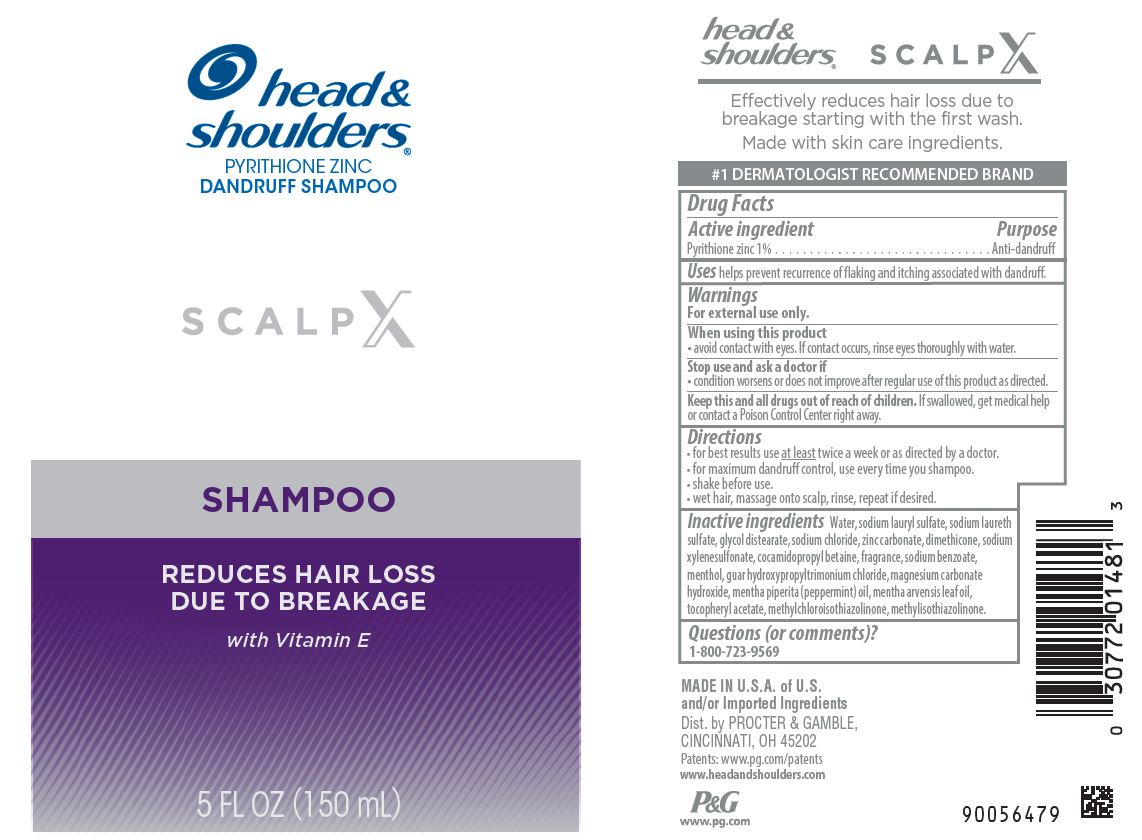
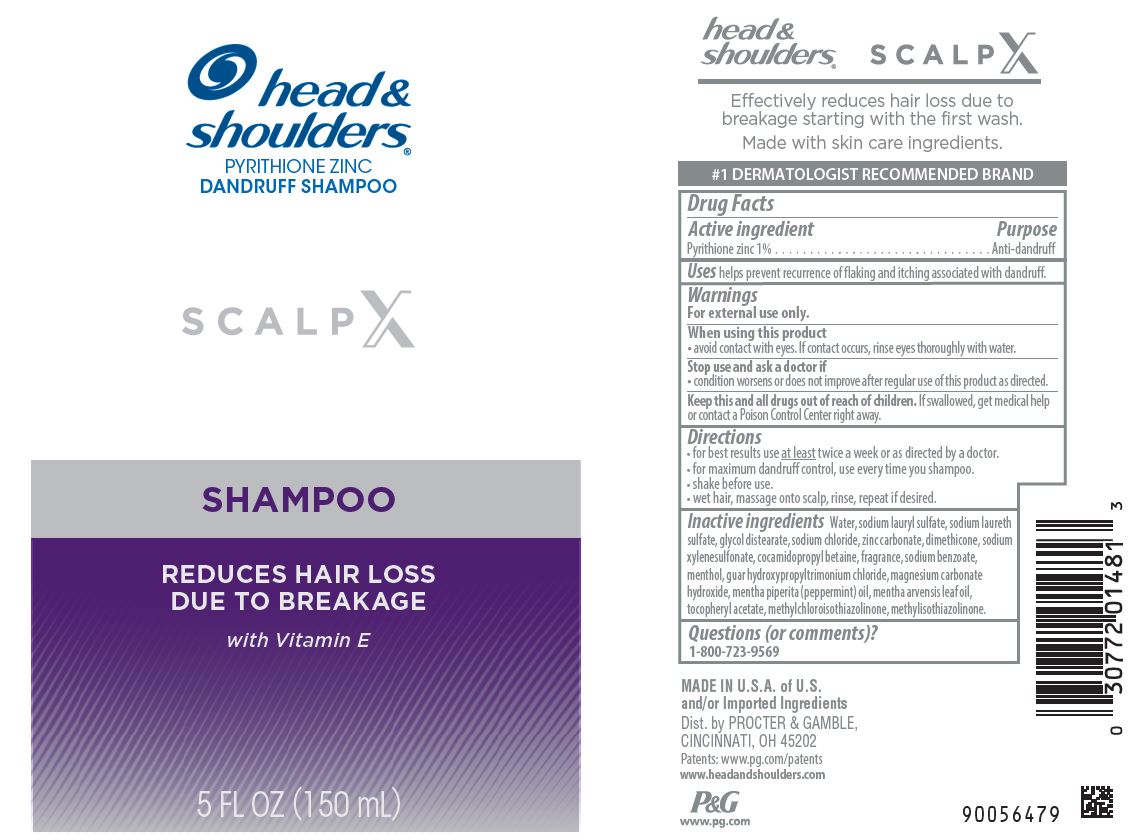
Label: HEAD AND SHOULDERS SCALP X- pyrithione zinc shampoo
- NDC Code(s): 69423-643-15
- Packager: The Procter & Gamble Manufacturing Company
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated December 15, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
- Warnings
- Directions
-
Inactive Ingredients
Water, sodium lauryl sulfate, sodium laureth sulfate, glycol distearate, sodium chloride, zinc carbonate, dimethicone, sodium xylenesulfonate, cocamidopropyl betaine, fragrance, sodium benzoate, menthol, guar hydroxypropyltrimonium chloride, magnesium carbonate hydroxide, mentha piperita (peppermint) oil, mentha arvensis leaf oil, tocopheryl acetate, methylchloroisothiazolinone, methylisothiazolinone
- Questions (or comments)?
- SPL UNCLASSIFIED SECTION
- Principal display panel - 150 mL Bottle Label
-
INGREDIENTS AND APPEARANCE
HEAD AND SHOULDERS SCALP X
pyrithione zinc shampooProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69423-643 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PYRITHIONE ZINC (UNII: R953O2RHZ5) (PYRITHIONE ZINC - UNII:R953O2RHZ5) PYRITHIONE ZINC 1 g in 100 mL Inactive Ingredients Ingredient Name Strength METHYLCHLOROISOTHIAZOLINONE (UNII: DEL7T5QRPN) DIMETHICONE (UNII: 92RU3N3Y1O) GLYCOL DISTEARATE (UNII: 13W7MDN21W) SODIUM CHLORIDE (UNII: 451W47IQ8X) ALPHA-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) SODIUM XYLENESULFONATE (UNII: G4LZF950UR) METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) GUAR HYDROXYPROPYLTRIMONIUM CHLORIDE (1.7 SUBSTITUENTS PER SACCHARIDE) (UNII: B16G315W7A) MAGNESIUM CARBONATE HYDROXIDE (UNII: YQO029V1L4) PEPPERMINT OIL (UNII: AV092KU4JH) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) SODIUM LAURYL SULFATE (UNII: 368GB5141J) WATER (UNII: 059QF0KO0R) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM LAURETH SULFATE (UNII: BPV390UAP0) MENTHA ARVENSIS LEAF OIL (UNII: 1AEY1M553N) ZINC CARBONATE (UNII: EQR32Y7H0M) MENTHOL (UNII: L7T10EIP3A) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69423-643-15 150 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 09/01/2022 09/01/2024 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M032 09/01/2022 09/01/2024 Labeler - The Procter & Gamble Manufacturing Company (004238200) Establishment Name Address ID/FEI Business Operations The Procter & Gamble Manufacturing Company 081329183 manufacture(69423-643) , pack(69423-643)