Label: PROMACE- acepromazine maleate tablet
- NDC Code(s): 0010-6704-01, 0010-6704-02
- Packager: Boehringer Ingelheim Animal Health USA Inc.
- Category: PRESCRIPTION ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Animal Drug Application
Drug Label Information
Updated December 17, 2021
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Caution:
- Description:
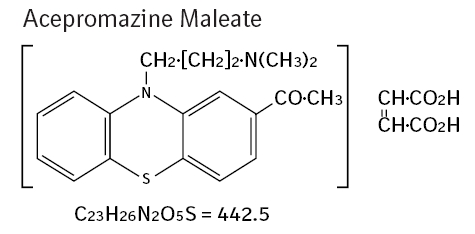
- Chemistry:
-
Mode of Action:
Acepromazine maleate has a depressant effect on the central nervous system and therefore causes sedation, muscular relaxation and a reduction in spontaneous activity. It acts rapidly, exerting a prompt and pronounced calming effect. It is an effective preanesthetic agent and lowers the dosage requirement of general anesthetics.
- Indications:
- Dosage and Administration:
-
Animal Safety:
Acute and chronic toxicity studies have shown a very low order of toxicity for acepromazine maleate.
A safety study using elevated dosages of acepromazine maleate demonstrated no adverse reactions even when administered at three times the upper limit of the recommended daily dosage (3.0 mg/lb body weight). The clinical observation for this high dosage was mild depression which disappeared in most dogs 24 hours after termination of dosing.
The only occurrence of adverse reaction during numerous clinical trials was a very mild respiratory distress (reverse sneeze) which was transient in nature and had no effect on the desired action of the drug.
-
Contraindications:
Phenothiazines may potentiate the toxicity of organophosphates. Therefore, do not use acepromazine maleate to control tremors associated with organic phosphate poisoning.
Do not use in conjunction with organophosphorus vermifuges or ectoparasiticides, including flea collars.
Do not use with procaine hydrochloride.
- Warning:
-
Precautions:
Tranquilizers are potent central nervous system depressants, and they can cause marked sedation with suppression of the sympathetic nervous system. Tranquilizers can produce prolonged depression or motor restlessness when given in excessive amounts or when given to sensitive animals.
Tranquilizers are additive in action to the actions of other depressants and will potentiate general anesthesia. Tranquilizers should be administered in smaller doses and with greater care during general anesthesia and also to animals exhibiting symptoms of stress, debilitation, cardiac disease, sympathetic blockade, hypovolemia or shock. Acepromazine, like other phenothiazine derivatives, is detoxified in the liver; therefore, it should be used with caution on animals with a previous history of liver dysfunction or leukopenia.
Epinephrine is contraindicated for treatment of acute hypotension produced by phenothiazine-derivative tranquilizers since further depression of blood pressure can occur.
Phenothiazines should be used with caution when followed by epidural anesthetic procedures because they may potentiate the arterial hypotensive effects of local anesthetics.
-
Adverse Reactions:
A few rare but serious occurrences of idiosyncratic reactions to acepromazine may occur in dogs following oral or parenteral administration. These potentially serious adverse reactions include behavioral disorders in dogs such as aggression, biting/chewing, and nervousness.
To report suspected adverse events, for technical assistance, or to obtain a copy of the Safety Data Sheet, contact Boehringer Ingelheim Animal Health USA Inc. at 1-888-637-4251.
For additional information about adverse drug experience reporting for animal drugs, contact FDA by telephone at 1-888-FDA-VETS or online at www.fda.gov/reportanimalae.
- Storage:
- How Supplied:
- SPL UNCLASSIFIED SECTION
- Principle Display Panel - Container Label, 25 mg, 500 count
-
INGREDIENTS AND APPEARANCE
PROMACE
acepromazine maleate tabletProduct Information Product Type PRESCRIPTION ANIMAL DRUG Item Code (Source) NDC:0010-6704 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACEPROMAZINE MALEATE (UNII: 37862HP2OM) (ACEPROMAZINE - UNII:54EJ303F0R) ACEPROMAZINE MALEATE 25 mg Inactive Ingredients Ingredient Name Strength MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) STARCH, CORN (UNII: O8232NY3SJ) MAGNESIUM STEARATE (UNII: 70097M6I30) DEXTROSE MONOHYDRATE (UNII: LX22YL083G) Product Characteristics Color YELLOW Score 4 pieces Shape ROUND Size 8mm Flavor Imprint Code 0221 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0010-6704-01 100 in 1 BOTTLE, PLASTIC 2 NDC:0010-6704-02 500 in 1 BOTTLE, PLASTIC Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NADA NADA117532 02/13/2013 Labeler - Boehringer Ingelheim Animal Health USA Inc. (007134091)