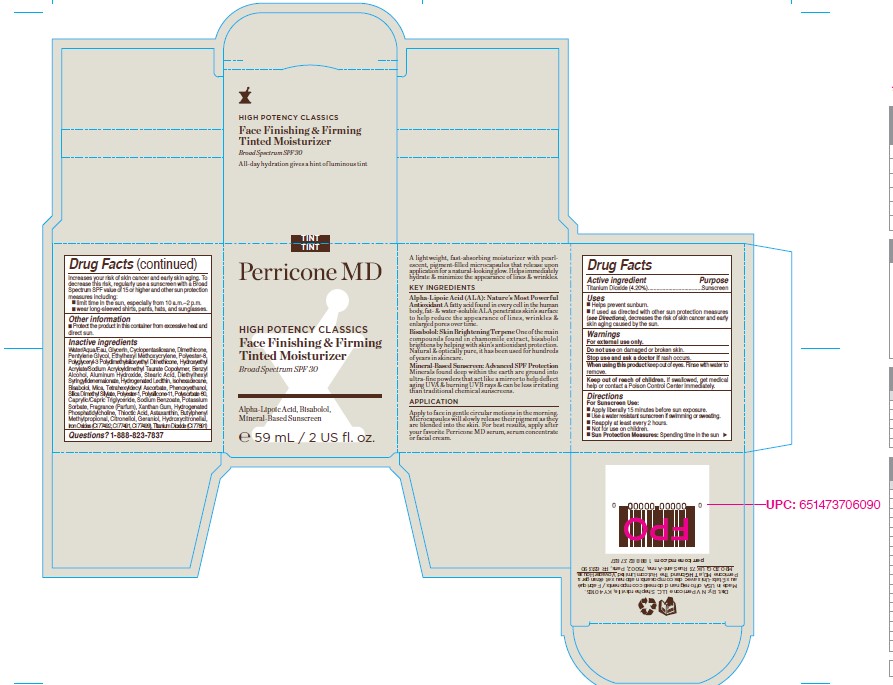
Label: HIGH POTENCY CLASSICS FACE FINISHING AND FIRMING TINTED MOISTURIZER SPF 30- titanium dioxide cream
- NDC Code(s): 45634-137-20
- Packager: N.V Perricone LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 10, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- DOSAGE & ADMINISTRATION
- ACTIVE INGREDIENT
- INACTIVE INGREDIENT
- INDICATIONS & USAGE
- KEEP OUT OF REACH OF CHILDREN
- PURPOSE
- WARNINGS
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
HIGH POTENCY CLASSICS FACE FINISHING AND FIRMING TINTED MOISTURIZER SPF 30
titanium dioxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:45634-137 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 2.478 g in 59 g Inactive Ingredients Ingredient Name Strength ISOHEXADECANE (UNII: 918X1OUF1E) DIMETHICONE CROSSPOLYMER (450000 MPA.S AT 12% IN CYCLOPENTASILOXANE) (UNII: UF7620L1W6) POLYESTER-8 (1400 MW, CYANODIPHENYLPROPENOYL CAPPED) (UNII: T9296U138P) HYDROXYETHYL ACRYLATE/SODIUM ACRYLOYLDIMETHYL TAURATE COPOLYMER (45000 MPA.S AT 1%) (UNII: 86FQE96TZ4) STEARIC ACID (UNII: 4ELV7Z65AP) TETRAHEXYLDECYL ASCORBATE (UNII: 9LBV3F07AZ) PHENOXYETHANOL (UNII: HIE492ZZ3T) SORBITAN ISOSTEARATE (UNII: 01S2G2C1E4) SODIUM BENZOATE (UNII: OJ245FE5EU) XANTHAN GUM (UNII: TTV12P4NEE) HYDROGENATED SOYBEAN PHOSPHATIDYLCHOLINE (UNII: QHQ97HF0PY) THIOCTIC ACID (UNII: 73Y7P0K73Y) PENTYLENE GLYCOL (UNII: 50C1307PZG) POLYSORBATE 60 (UNII: CAL22UVI4M) ALUMINUM HYDROXIDE (UNII: 5QB0T2IUN0) DIETHYLHEXYL SYRINGYLIDENEMALONATE (UNII: 3V5U97P248) HYDROGENATED SOYBEAN LECITHIN (UNII: H1109Z9J4N) MANNITOL (UNII: 3OWL53L36A) .BETA.-BISABOLOL (UNII: LP618AV2EA) MEDIUM-CHAIN TRIGLYCERIDES (UNII: C9H2L21V7U) ACETYL TETRAPEPTIDE-9 (UNII: VMO8OOD3V0) WATER (UNII: 059QF0KO0R) GLYCERIN (UNII: PDC6A3C0OX) BENZYL ALCOHOL (UNII: LKG8494WBH) POLYSILICONE-15 (UNII: F8DRP5BB29) POLYGLYCERYL-3 POLYDIMETHYLSILOXYETHYL DIMETHICONE (4000 MPA.S) (UNII: RLA2U05Z4Q) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) NIACINAMIDE (UNII: 25X51I8RD4) ETHYLHEXYL METHOXYCRYLENE (UNII: S3KFG6Q5X8) Product Characteristics Color yellow Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:45634-137-20 82 g in 1 BOTTLE, GLASS; Type 0: Not a Combination Product 04/04/2022 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final M020 04/04/2022 Labeler - N.V Perricone LLC (054414243) Establishment Name Address ID/FEI Business Operations Dimensional Merchandising Inc (DMI) 076693183 manufacture(45634-137)