Label: EQUATE PAIN RELIEF BALM- menthol 11%, camphor 11% ointment ointment
- NDC Code(s): 79903-105-18
- Packager: WALMART, INC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated March 15, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS
- PURPOSE
- USES
-
WARNINGS
For external use only.
When using this product
- use only as directed.
- avoid contact with eye and mucous membranes
- do not apply to wounds, damaged or irritated skin
- do not bandage or cover with wrap or heating pad
- do not use 1 hour prior to bathing or within 30 minutes after bathing
Stop use an ask a doctor if
- condition worsens
- severe skin irritation occurs
- pain persists for more than 7 days
- pain clears up and then recurs a few days later
- DIRECTIONS
- OTHER INFORMATION
- INACTIVE INGREDIENTS
- QUESTIONS OR COMMENTS
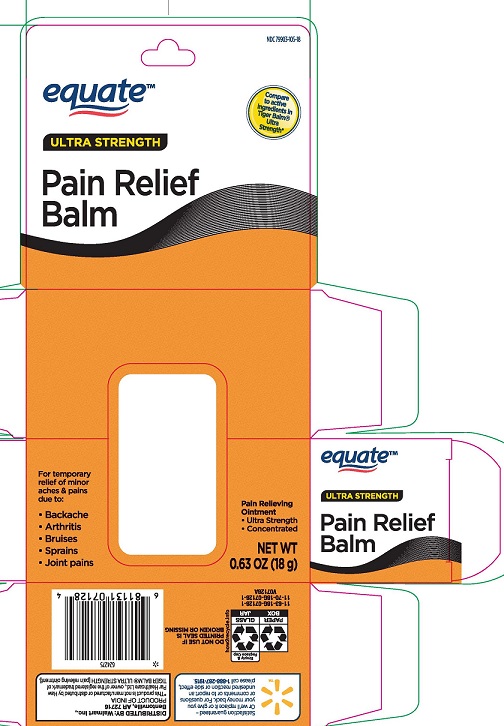
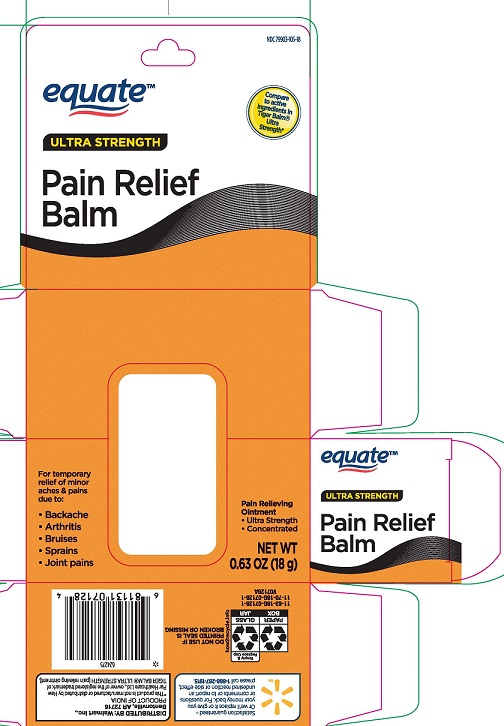
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
EQUATE PAIN RELIEF BALM
menthol 11%, camphor 11% ointment ointmentProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79903-105 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MENTHOL (UNII: L7T10EIP3A) (MENTHOL - UNII:L7T10EIP3A) MENTHOL 110 mg in 1 g CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) (CAMPHOR (SYNTHETIC) - UNII:5TJD82A1ET) CAMPHOR (SYNTHETIC) 110 mg in 1 g Inactive Ingredients Ingredient Name Strength CAJUPUT OIL (UNII: J3TO6BUQ37) CLOVE OIL (UNII: 578389D6D0) CHINESE CINNAMON OIL (UNII: A4WO0626T5) PARAFFIN (UNII: I9O0E3H2ZE) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79903-105-18 1 in 1 CARTON 04/01/2022 1 18 g in 1 BOTTLE, GLASS; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M017 04/01/2022 Labeler - WALMART, INC (051957769)