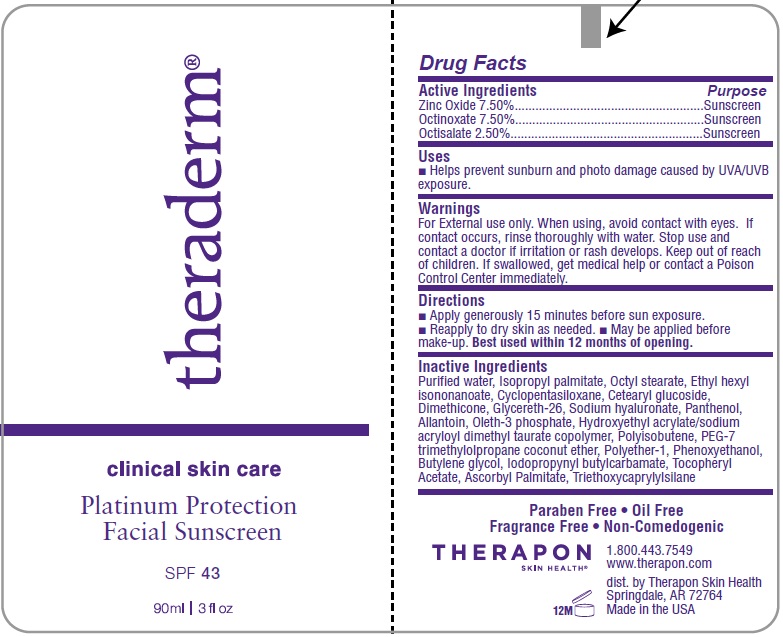
Label: THERADERMSPF 43 SPF 43- zinc oxide, octinoxate, octisalate emulsion
-
Contains inactivated NDC Code(s)
NDC Code(s): 66428-002-02 - Packager: Therapon Skin Health, LP
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 20, 2012
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Uses
- Warnings
- Directions
-
Inactive Ingredients
Purified water, Isopropyl palmitate, Octyl stearate, Ethyl hexyl isononanoate,Cyclopentasiloxane, Cetearyl glucoside, Dimethicone, Glycereth-26, Sodium Hyaluronate, Panthenol, Allantoin, Oleth-3 phosphate, Hydroxyethyl Acrylate/Sodium acryloyl dimethyl taurate copolymer, Polyisobutene, PEG-7 trimethylolpropane coconut ether, Polyether-1, Phenoxyethanol, Butylene glycol, Iodopropynyl butylcarbamate, Tocopheryl acetate, Triethoxycaprylylsilane.
- Label for Tube and Box
-
INGREDIENTS AND APPEARANCE
THERADERMSPF 43 SPF 43
zinc oxide, octinoxate, octisalate emulsionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:66428-002 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Zinc Oxide (UNII: SOI2LOH54Z) (Zinc Oxide - UNII:SOI2LOH54Z) Zinc Oxide 7.5 mL in 100 mL Octinoxate (UNII: 4Y5P7MUD51) (Octinoxate - UNII:4Y5P7MUD51) Octinoxate 7.5 mL in 100 mL Octisalate (UNII: 4X49Y0596W) (Octisalate - UNII:4X49Y0596W) Octisalate 2.5 mL in 100 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Isopropyl Palmitate (UNII: 8CRQ2TH63M) Octyl Stearate (UNII: 772Y4UFC8B) Ethylhexyl Isononanoate (UNII: I6KB4GE3K4) Cyclomethicone 5 (UNII: 0THT5PCI0R) Cetearyl Glucoside (UNII: 09FUA47KNA) Dimethicone (UNII: 92RU3N3Y1O) Glycereth-26 (UNII: NNE56F2N14) Hyaluronate Sodium (UNII: YSE9PPT4TH) Panthenol (UNII: WV9CM0O67Z) Allantoin (UNII: 344S277G0Z) Oleth-3 Phosphate (UNII: 8Q0Z18J1VL) 2-Hydroxyethyl Acrylate (UNII: 25GT92NY0C) Polyisobutylene (1300 MW) (UNII: 241BN7J12Y) Coconut Alcohol (UNII: 13F4MW8Y9K) Polyester-7 (UNII: 0841698D2F) Ethylene Oxide (UNII: JJH7GNN18P) Polyethylene Glycol 350 (UNII: ZBR3T82M2V) Polyisobutylene (1300 MW) (UNII: 241BN7J12Y) Phenoxyethanol (UNII: HIE492ZZ3T) Butylene Glycol (UNII: 3XUS85K0RA) Iodopropynyl Butylcarbamate (UNII: 603P14DHEB) .Alpha.-Tocopherol Acetate (UNII: 9E8X80D2L0) Ascorbyl Palmitate (UNII: QN83US2B0N) Triethoxycaprylylsilane (UNII: LDC331P08E) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:66428-002-02 1 in 1 BOX 1 90 mL in 1 TUBE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 01/20/2012 Labeler - Therapon Skin Health, LP (792518995) Establishment Name Address ID/FEI Business Operations Swiss American Products, Inc. 611921669 manufacture