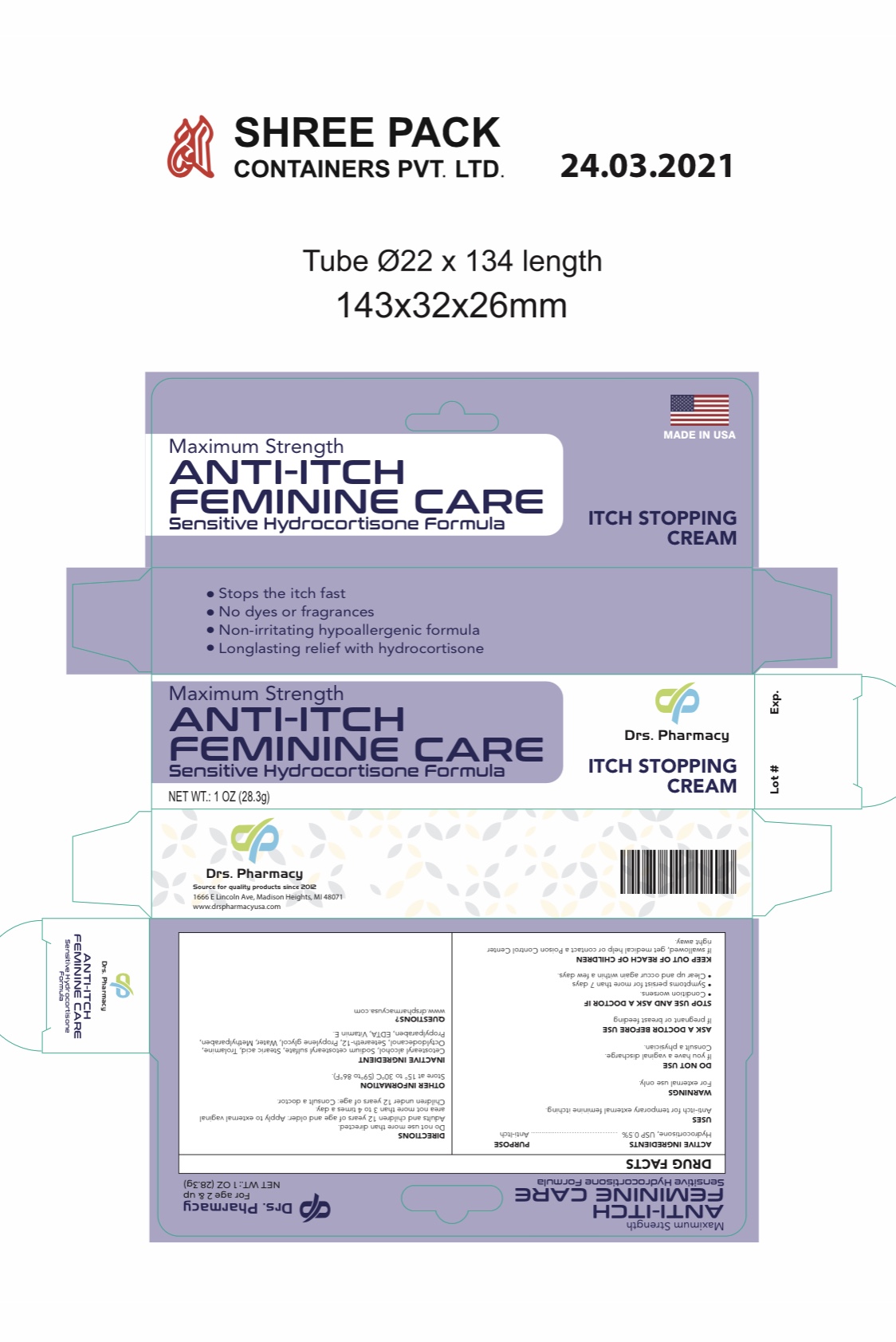
Label: ANTI-ITCH FEMININE CARE- hydrocortisone 0.5% cream
- NDC Code(s): 80489-103-01, 80489-103-02
- Packager: OL PHARMA TECH, LLC Drs PHARMACY
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 8, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT
- PURPOSE
- USES
- WARNINGS
- DIRETIONS
- Stop use and ask a doctor if
- KEEP OUT OF REACH OF CHILDREN
- OTHER INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
- PACKAGE LABEL
-
INGREDIENTS AND APPEARANCE
ANTI-ITCH FEMININE CARE
hydrocortisone 0.5% creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:80489-103 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HYDROCORTISONE (UNII: WI4X0X7BPJ) (HYDROCORTISONE - UNII:WI4X0X7BPJ) HYDROCORTISONE 5 mg in 1 g Inactive Ingredients Ingredient Name Strength PROPYLENE GLYCOL (UNII: 6DC9Q167V3) PROPYLPARABEN (UNII: Z8IX2SC1OH) .ALPHA.-TOCOPHEROL (UNII: H4N855PNZ1) METHYLPARABEN (UNII: A2I8C7HI9T) EDETATE DISODIUM (UNII: 7FLD91C86K) CETEARETH-12 (UNII: 7V4MR24V5P) WATER (UNII: 059QF0KO0R) STEARIC ACID (UNII: 4ELV7Z65AP) SODIUM CETOSTEARYL SULFATE (UNII: 7ZBS06BH4B) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) OCTYLDODECANOL (UNII: 461N1O614Y) TROLAMINE (UNII: 9O3K93S3TK) Product Characteristics Color white Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:80489-103-01 1 in 1 CARTON 02/01/2022 1 20 g in 1 TUBE; Type 0: Not a Combination Product 2 NDC:80489-103-02 1 in 1 CARTON 02/01/2022 2 30 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M005 02/01/2022 Labeler - OL PHARMA TECH, LLC Drs PHARMACY (021170377) Registrant - OL PHARMA TECH, LLC Drs PHARMACY (021170377) Establishment Name Address ID/FEI Business Operations OHIO LAB PHARMA 021170377 manufacture(80489-103)