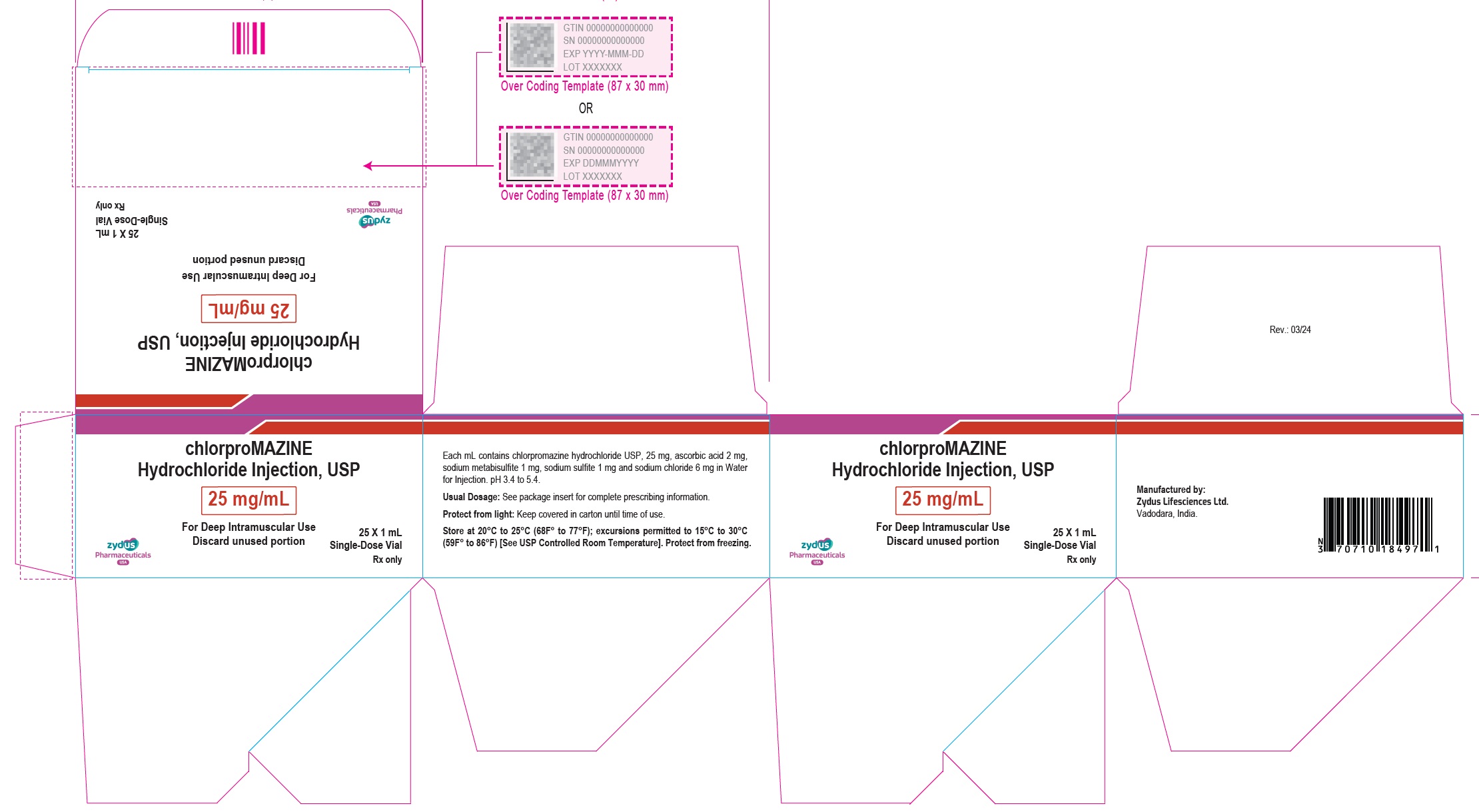
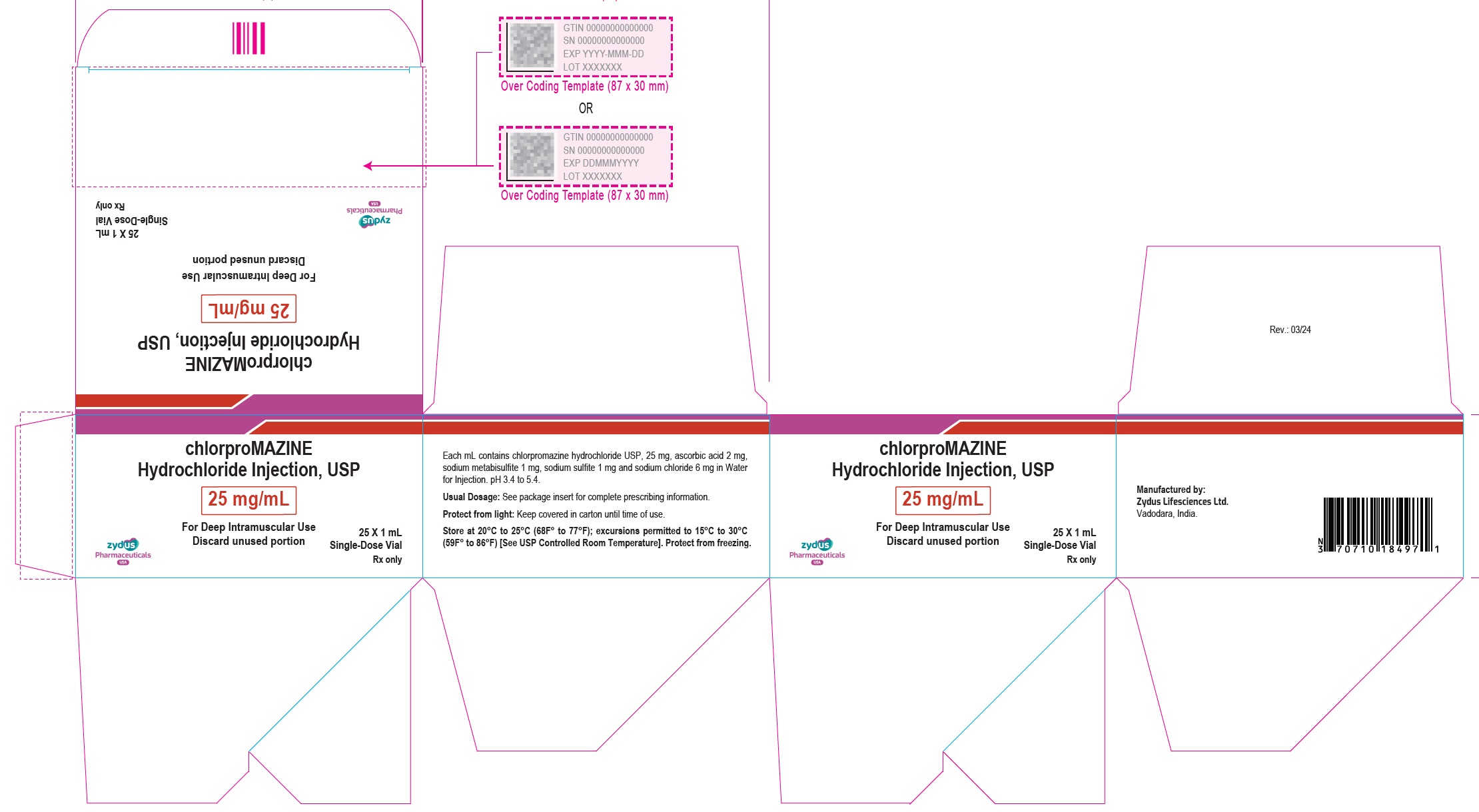
Label: CHLORPROMAZINE HYDROCHLORIDE injection
- NDC Code(s): 70771-1778-1, 70771-1778-7, 70771-1779-1, 70771-1779-7
- Packager: Zydus Lifesciences Limited
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated March 14, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CHLORPROMAZINE HYDROCHLORIDE
chlorpromazine hydrochloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1779 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORPROMAZINE HYDROCHLORIDE (UNII: 9WP59609J6) (CHLORPROMAZINE - UNII:U42B7VYA4P) CHLORPROMAZINE HYDROCHLORIDE 25 mg in 1 mL Inactive Ingredients Ingredient Name Strength ASCORBIC ACID (UNII: PQ6CK8PD0R) 2 mg in 1 mL SODIUM METABISULFITE (UNII: 4VON5FNS3C) 1 mg in 1 mL SODIUM SULFITE (UNII: VTK01UQK3G) 1 mg in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 6 mg in 1 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1779-7 25 in 1 CARTON 03/29/2024 1 NDC:70771-1779-1 2 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA217275 03/29/2024 CHLORPROMAZINE HYDROCHLORIDE
chlorpromazine hydrochloride injectionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:70771-1778 Route of Administration INTRAMUSCULAR Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CHLORPROMAZINE HYDROCHLORIDE (UNII: 9WP59609J6) (CHLORPROMAZINE - UNII:U42B7VYA4P) CHLORPROMAZINE HYDROCHLORIDE 25 mg in 1 mL Inactive Ingredients Ingredient Name Strength ASCORBIC ACID (UNII: PQ6CK8PD0R) 2 mg in 1 mL SODIUM METABISULFITE (UNII: 4VON5FNS3C) 1 mg in 1 mL SODIUM SULFITE (UNII: VTK01UQK3G) 1 mg in 1 mL SODIUM CHLORIDE (UNII: 451W47IQ8X) 6 mg in 1 mL WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:70771-1778-7 25 in 1 CARTON 03/29/2024 1 NDC:70771-1778-1 1 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA217275 03/29/2024 Labeler - Zydus Lifesciences Limited (918596198) Registrant - Zydus Pharmaceuticals USA Inc. (156861945) Establishment Name Address ID/FEI Business Operations Zydus Lifesciences Limited 873671928 MANUFACTURE(70771-1778, 70771-1779) , ANALYSIS(70771-1778, 70771-1779)