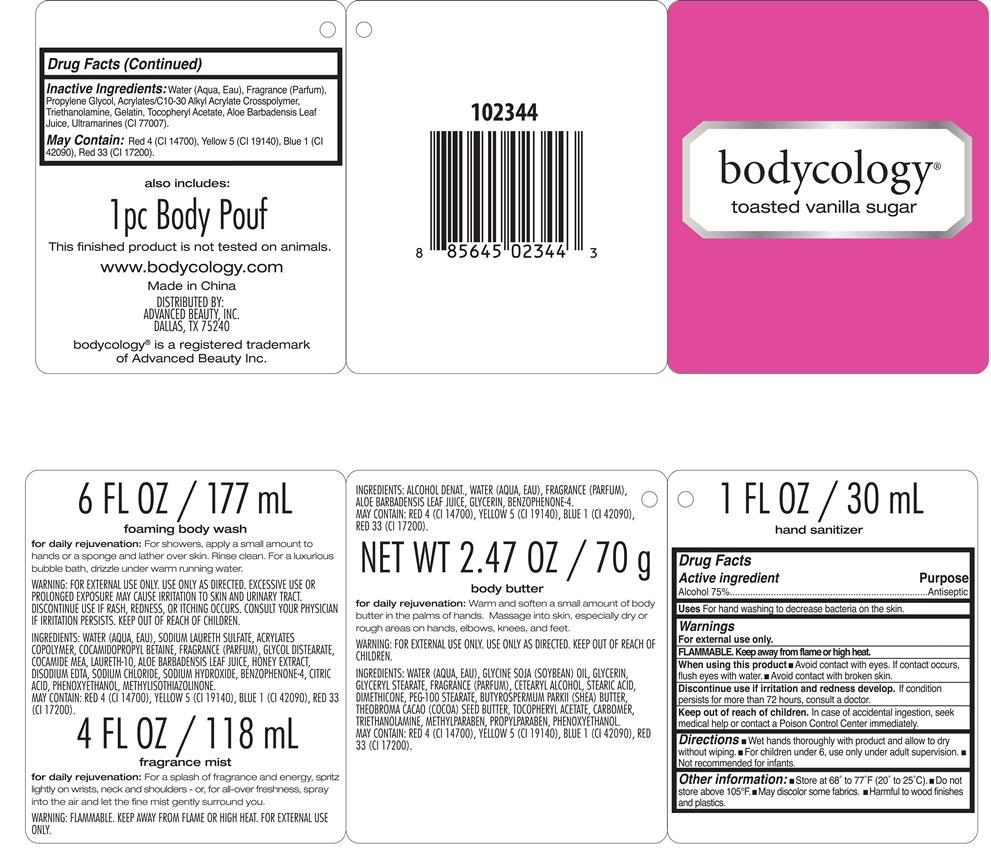
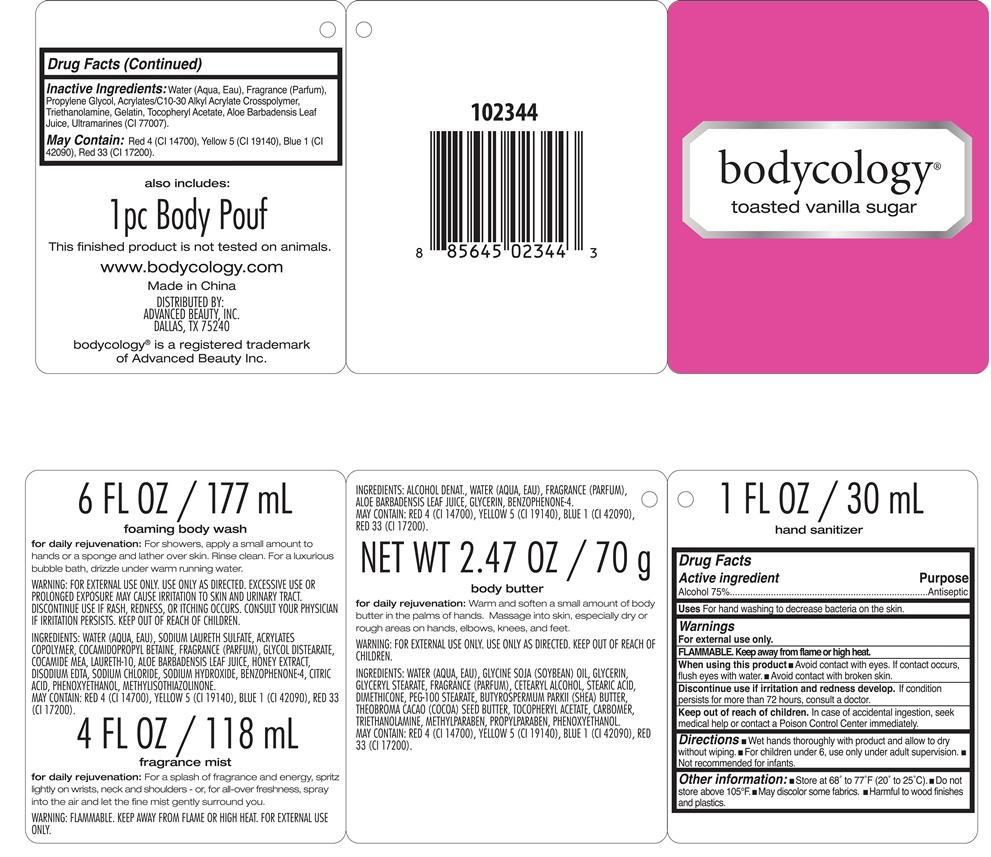
Label: TOASTED VANILLA SUGAR KIT BODYCOLOGY- alcohol kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 51345-109-01 - Packager: Advanced Beauty Systems, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 2, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active Ingredient Purpose
Alcohol 75% Antiseptic
Uses For hand washing to decrease bacteria on the skinKeep out of reach of children. In case of accidental ingestion, seek medical help or contact a Poison Control Center immediately.
Discontinue use if irritation and redness develop. If condition persists for more than 72 hours, consult a doctor.
Warnings
For external use only
Flammable. Keep away from flame or high heat.
When using this product
- Avoid contact with eyes. If contact occurs flush eyes with water.
- Avoid contact with Broken skin
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
TOASTED VANILLA SUGAR KIT BODYCOLOGY
alcohol kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:51345-109 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51345-109-01 1 in 1 KIT Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE 30 mL Part 2 1 BOTTLE 177 mL Part 3 1 BOTTLE, SPRAY 118 mL Part 4 1 JAR 70 g Part 1 of 4 TOASTED VANILLA SUGAR HAND SANITIZER BODYCOLOGY
alcohol liquidProduct Information Item Code (Source) NDC:51345-110 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 75 g in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) CARBOMER COPOLYMER TYPE A (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: 71DD5V995L) TROLAMINE (UNII: 9O3K93S3TK) GELATIN (UNII: 2G86QN327L) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) ALOE VERA LEAF (UNII: ZY81Z83H0X) ULTRAMARINE BLUE (UNII: I39WR998BI) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 30 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 06/25/2013 Part 2 of 4 TOASTED VANILLA SUGAR FOAMING BODY WASH
body and hand (excluding shaving preparations)Product Information Other Ingredients Ingredient Kind Ingredient Name Quantity INGR WATER (UNII: 059QF0KO0R) INGR SODIUM LAURETH SULFATE (UNII: BPV390UAP0) INGR COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) INGR GLYCOL DISTEARATE (UNII: 13W7MDN21W) INGR COCO MONOETHANOLAMIDE (UNII: C80684146D) INGR LAURETH-10 (UNII: BD7AST04GA) INGR ALOE VERA LEAF (UNII: ZY81Z83H0X) INGR HONEY (UNII: Y9H1V576FH) INGR EDETATE DISODIUM (UNII: 7FLD91C86K) INGR SODIUM CHLORIDE (UNII: 451W47IQ8X) INGR SODIUM HYDROXIDE (UNII: 55X04QC32I) INGR SULISOBENZONE (UNII: 1W6L629B4K) INGR CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) INGR PHENOXYETHANOL (UNII: HIE492ZZ3T) INGR METHYLISOTHIAZOLINONE (UNII: 229D0E1QFA) INGR FD&C RED NO. 4 (UNII: X3W0AM1JLX) INGR FD&C YELLOW NO. 5 (UNII: I753WB2F1M) INGR FD&C BLUE NO. 1 (UNII: H3R47K3TBD) INGR D&C RED NO. 33 (UNII: 9DBA0SBB0L) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 177 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Cosmetic 06/25/2013 Part 3 of 4 TOASTED VANILLA SUGAR FRAGRANCE MIST
other fragrance preparationsProduct Information Other Ingredients Ingredient Kind Ingredient Name Quantity INGR WATER (UNII: 059QF0KO0R) INGR ALOE VERA LEAF (UNII: ZY81Z83H0X) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR SULISOBENZONE (UNII: 1W6L629B4K) INGR FD&C RED NO. 4 (UNII: X3W0AM1JLX) INGR FD&C YELLOW NO. 5 (UNII: I753WB2F1M) INGR FD&C BLUE NO. 1 (UNII: H3R47K3TBD) INGR D&C RED NO. 33 (UNII: 9DBA0SBB0L) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 118 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Cosmetic 06/25/2013 Part 4 of 4 TOASTED VANILLA SUGAR BODY BUTTER
body and hand (excluding shaving preparations)Product Information Other Ingredients Ingredient Kind Ingredient Name Quantity INGR WATER (UNII: 059QF0KO0R) INGR SOYBEAN OIL (UNII: 241ATL177A) INGR GLYCERIN (UNII: PDC6A3C0OX) INGR GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) INGR CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) INGR STEARIC ACID (UNII: 4ELV7Z65AP) INGR DIMETHICONE (UNII: 92RU3N3Y1O) INGR PEG-100 STEARATE (UNII: YD01N1999R) INGR SHEA BUTTER (UNII: K49155WL9Y) INGR COCOA BUTTER (UNII: 512OYT1CRR) INGR .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) INGR CARBOMER 940 (UNII: 4Q93RCW27E) INGR TROLAMINE (UNII: 9O3K93S3TK) INGR METHYLPARABEN (UNII: A2I8C7HI9T) INGR PROPYLPARABEN (UNII: Z8IX2SC1OH) INGR PHENOXYETHANOL (UNII: HIE492ZZ3T) INGR FD&C RED NO. 4 (UNII: X3W0AM1JLX) INGR FD&C YELLOW NO. 5 (UNII: I753WB2F1M) INGR FD&C BLUE NO. 1 (UNII: H3R47K3TBD) INGR D&C RED NO. 33 (UNII: 9DBA0SBB0L) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 70 g in 1 JAR Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Cosmetic 06/25/2013 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333E 06/25/2013 Labeler - Advanced Beauty Systems, Inc. (129986613) Registrant - Advanced Beauty Systems, Inc. (129986613) Establishment Name Address ID/FEI Business Operations Landy International 545291775 manufacture(51345-109, 51345-110)