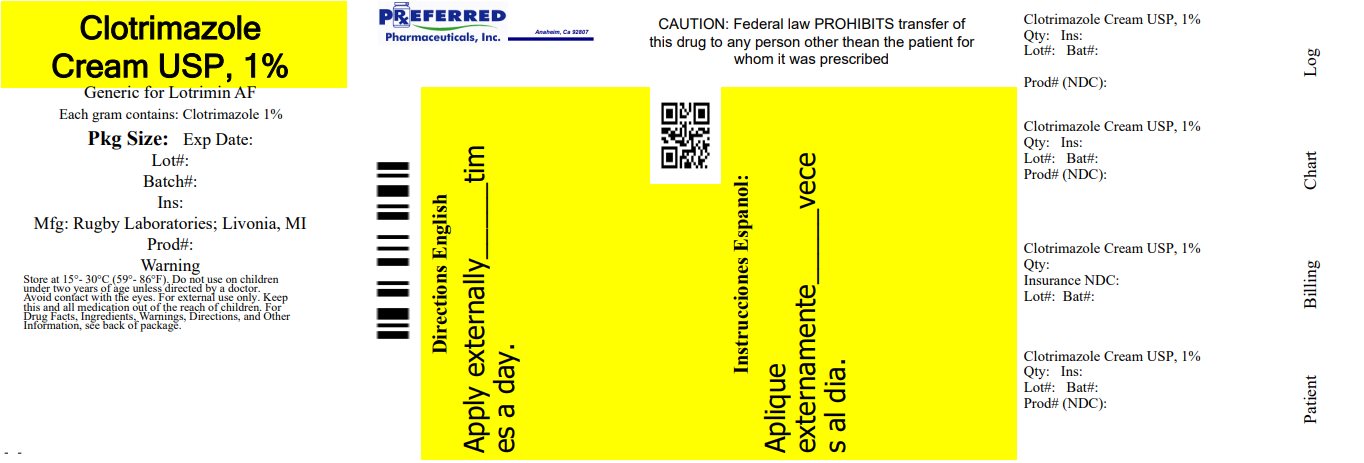
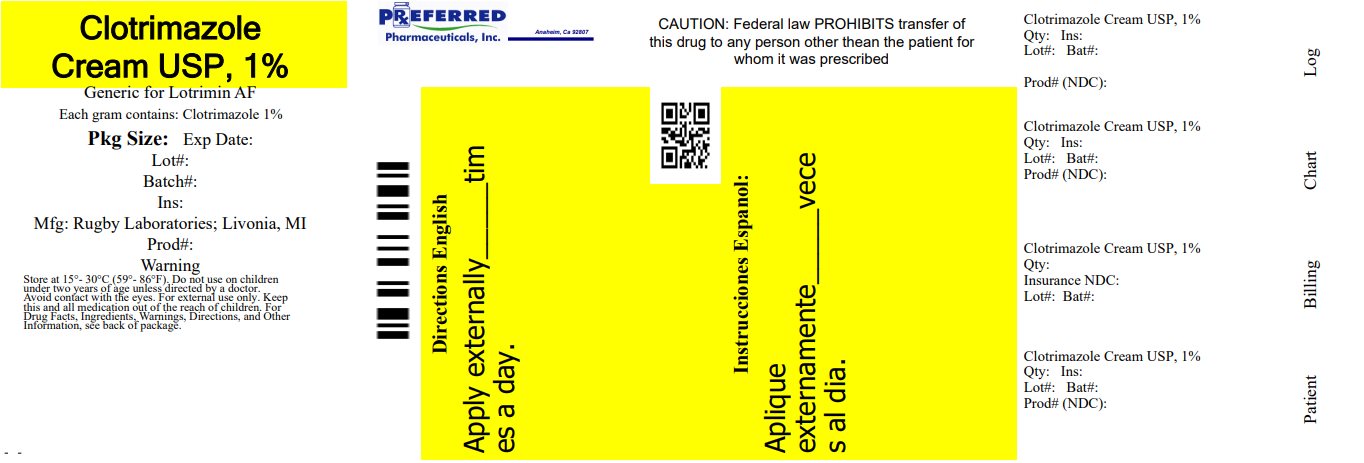
Label: RUGBY ANTIFUNGAL CLOTRIMAZOLE- clotrimazole cream
- NDC Code(s): 68788-8091-2
- Packager: Preferred Pharmaceuticals Inc.
- This is a repackaged label.
- Source NDC Code(s): 0536-1272
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 5, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
- Warnings
-
Directions
- •
- wash affected area and dry thoroughly
- •
- apply a thin layer over affected area twice daily (morning and night) or as directed by a doctor
- •
- supervise children in use of this product
- •
- for athlete's foot, pay special attention to spaces between the toes, wear well fitting, ventilated shoes and change shoes and socks at least once daily
- •
- for athlete's foot and ringworm, use daily for 4 weeks; for jock itch, use daily for 2 weeks
- •
- if condition persists longer, consult a doctor
- •
- this product is not effective on scalp or nails
- Other information
- Inactive ingredients
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 28 g Tube Carton
-
INGREDIENTS AND APPEARANCE
RUGBY ANTIFUNGAL CLOTRIMAZOLE
clotrimazole creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:68788-8091(NDC:0536-1272) Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CLOTRIMAZOLE (UNII: G07GZ97H65) (CLOTRIMAZOLE - UNII:G07GZ97H65) CLOTRIMAZOLE 0.01 g in 1 g Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) CETYL ALCOHOL (UNII: 936JST6JCN) DMDM HYDANTOIN (UNII: BYR0546TOW) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) ISOPROPYL ALCOHOL (UNII: ND2M416302) METHYLPARABEN (UNII: A2I8C7HI9T) MINERAL OIL (UNII: T5L8T28FGP) POLYSORBATE 20 (UNII: 7T1F30V5YH) PROPYLPARABEN (UNII: Z8IX2SC1OH) WATER (UNII: 059QF0KO0R) STEARIC ACID (UNII: 4ELV7Z65AP) TROLAMINE (UNII: 9O3K93S3TK) EDETATE TRISODIUM (UNII: 420IP921MB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:68788-8091-2 1 in 1 CARTON 10/05/2021 1 28 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug 333C 10/05/2021 Labeler - Preferred Pharmaceuticals Inc. (791119022) Registrant - Preferred Pharmaceuticals Inc. (791119022) Establishment Name Address ID/FEI Business Operations Preferred Pharmaceuticals Inc. 791119022 RELABEL(68788-8091)