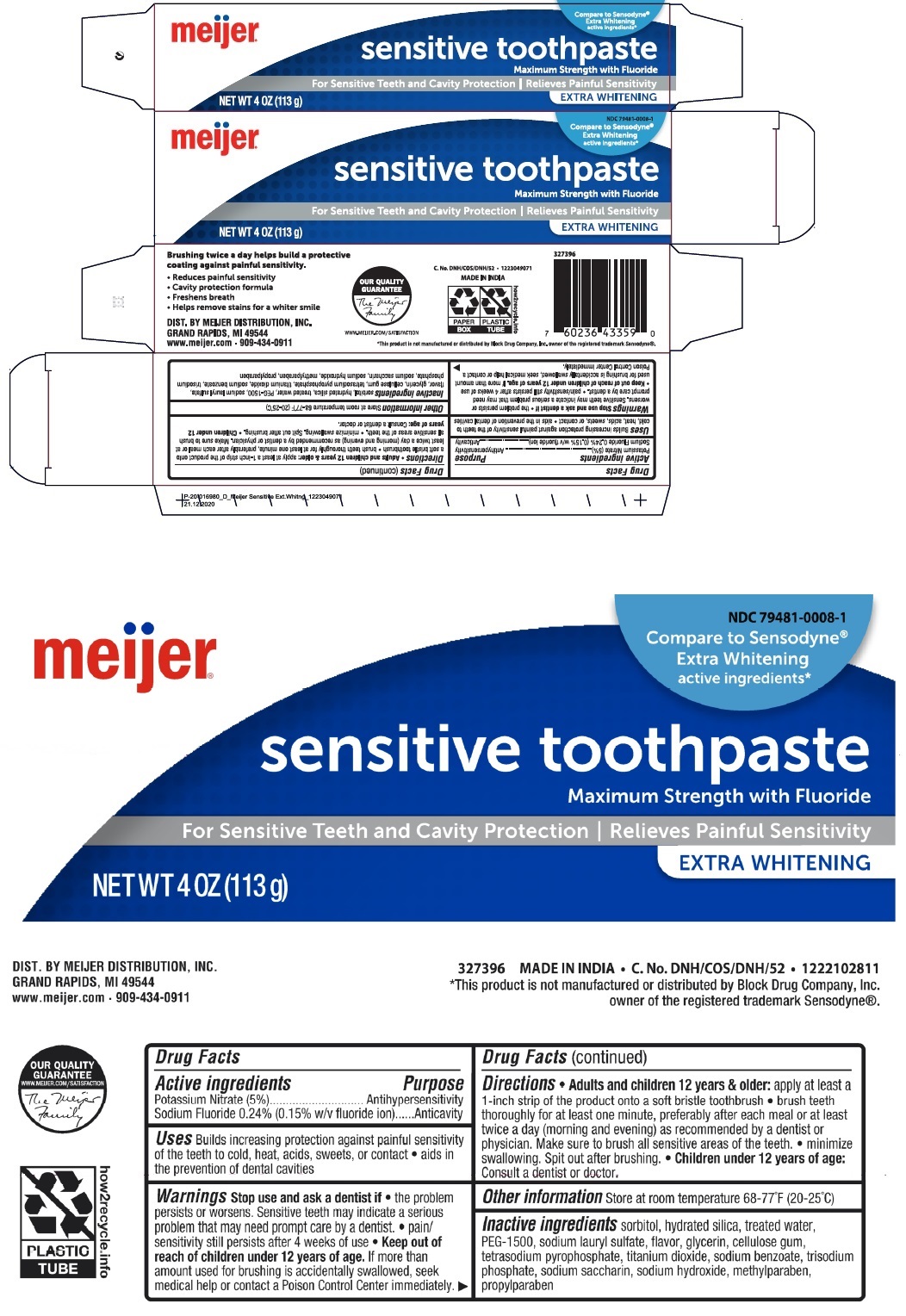
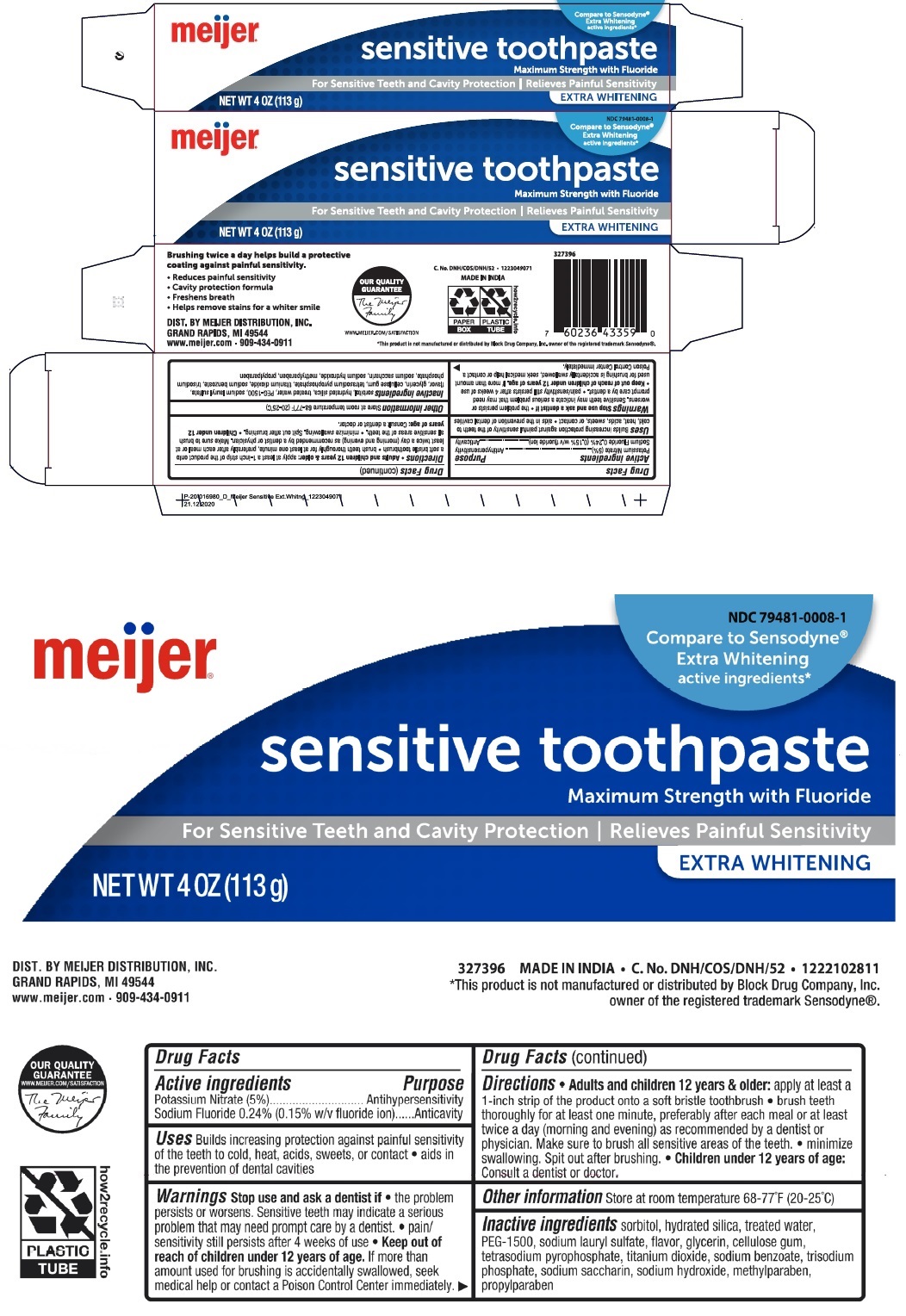
Label: SENSITIVE WHITENING TOOTH-PASTE- potassium nitrate, sodium fluoride paste, dentifrice
- NDC Code(s): 79481-0008-1
- Packager: Meijer, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated January 11, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredients
- Purpose
- Uses
- Warnings
-
Directions
• Adults and children 12 years & older: apply at least a 1-inch strip of the product onto a soft bristle toothbrush • brush teeth thoroughly for at least one minute, preferably after each meal or at least twice a day (morning and evening) as recommended by a dentist or physician. Make sure to brush all sensitive areas of the teeth. • minimize swallowing. Spit out after brushing. • Children under 12 years of age: Consult a dentist or doctor.
- Other information
- Inactive ingredients
-
SPL UNCLASSIFIED SECTION
Compare to Sensodyne®
Extra Whitening active ingredients*
Maximum Strength with Fluoride
For Sensitive Teeth and Cavity Protection | Relieves Painful Sensitivity
Brushing twice a day helps build a protective coating against painful sensitivity.
• Reduces painful sensitivity
• cavity protection formula
• Freshens breath
• Helps remove stains for a whiter smile
DIST. BY MEIJER DISTRIBUTION, INC.
GRAND RAPIDS, MI 49544
www.meijer.com • 909-434-0911
OUR QUALITY GUARANTEE
WWW.MEIJER.COM/SATISFACTION
MADE IN INDIA
PAPER BOX
PLASTIC TUBE
how2recycle.info
*This product is not manufactured or distributed by Block Drug Company, Inc. owner of the registered trademark Sensodyne®.
- Packaging
-
INGREDIENTS AND APPEARANCE
SENSITIVE WHITENING TOOTH-PASTE
potassium nitrate, sodium fluoride paste, dentifriceProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:79481-0008 Route of Administration DENTAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POTASSIUM NITRATE (UNII: RU45X2JN0Z) (NITRATE ION - UNII:T93E9Y2844) POTASSIUM NITRATE 50 mg in 1 g SODIUM FLUORIDE (UNII: 8ZYQ1474W7) (FLUORIDE ION - UNII:Q80VPU408O) FLUORIDE ION 2.43 mg in 1 g Inactive Ingredients Ingredient Name Strength SORBITOL (UNII: 506T60A25R) HYDRATED SILICA (UNII: Y6O7T4G8P9) WATER (UNII: 059QF0KO0R) POLYETHYLENE GLYCOL 1600 (UNII: 1212Z7S33A) SODIUM LAURYL SULFATE (UNII: 368GB5141J) GLYCERIN (UNII: PDC6A3C0OX) CARBOXYMETHYLCELLULOSE SODIUM, UNSPECIFIED (UNII: K679OBS311) SODIUM PYROPHOSPHATE (UNII: O352864B8Z) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) SODIUM BENZOATE (UNII: OJ245FE5EU) SODIUM PHOSPHATE, TRIBASIC, ANHYDROUS (UNII: SX01TZO3QZ) SACCHARIN SODIUM (UNII: SB8ZUX40TY) SODIUM HYDROXIDE (UNII: 55X04QC32I) METHYLPARABEN (UNII: A2I8C7HI9T) PROPYLPARABEN (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:79481-0008-1 1 in 1 BOX 12/24/2020 1 113 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 12/24/2020 Labeler - Meijer, Inc. (006959555)