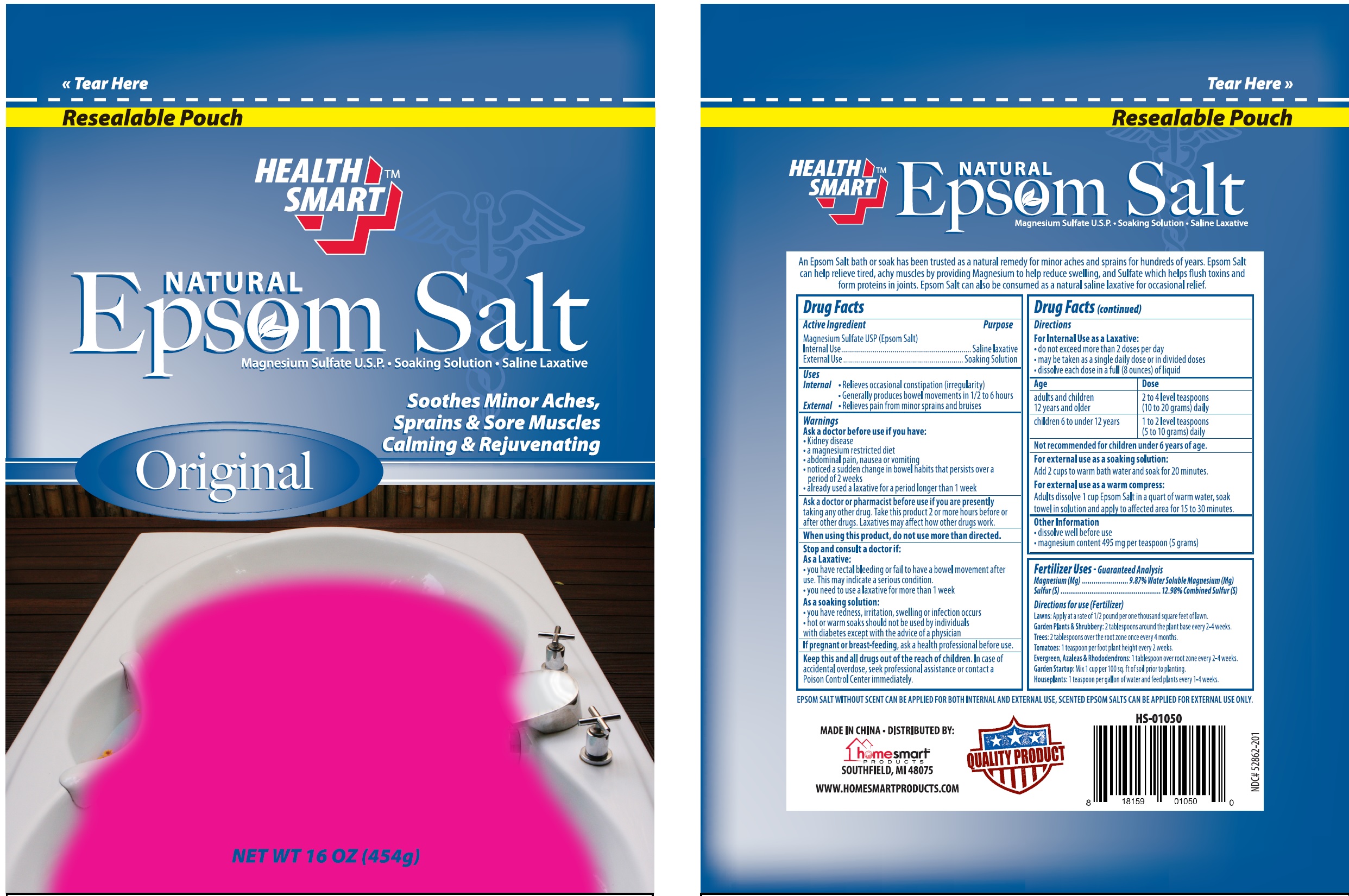
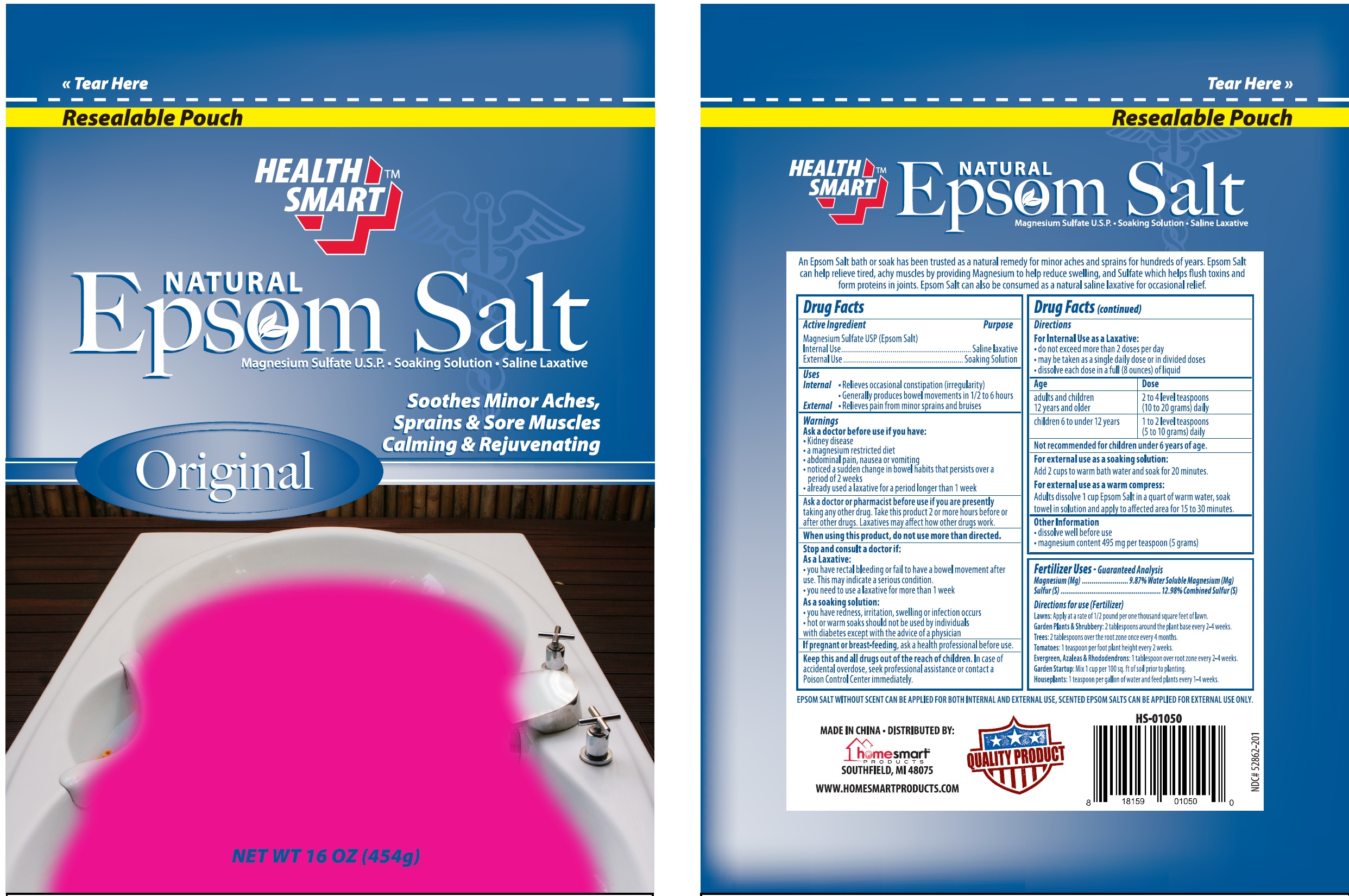
Label: HEALTH SMART NATURAL EPSOM SALT- magnesium sulfate granule, for solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 52862-201-16 - Packager: International Wholesale, Inc.
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated July 15, 2013
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have:
- Kidney disease
- a magnesium restricted diet
- abdominal pain, nausea or vomiting
- noticed a sudden change in bowel habbits that persists over a period of 2 weeks
- already used a laxative for a period longer than 1 week
Ask a doctor or pharmacist before use if you are presently taking any other drug. Take this product 2 or more hours before or after other drugs. Laxatives may affect how other drugs work.
- When using this product, do not use more than directed.
-
Stop and consult a doctor if:
As Laxative:
- you have rectal bleeding or fail to have a bowel movement after use. This may indicate a serious condition
- you need to use a laxative for more than 1 week
As soaking solution:
- you have redness, irritation, swelling or infection occurs
- hot or warm soaks should not be used by individuals with diabeties except with the advice of a physician
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
-
Directions
for Internal Use as a Laxative:
- do not exceed more than 2 doses per day
- may be taken as a single daily dose or in divided doses
- dissolve each dose in a full (8 ounces) of liquid
Age Dose adults and children 12 years and older 2 to 4 level teaspoons (10 to 20 grams) daily children 6 to under 12 years 1 to 2 level teaspoons (5 to 10 grams) daily Not recomended for children under 6 years of age
- DOSAGE & ADMINISTRATION
- Other Information
-
Fertilizer Uses - Guaranteed Analysis
Magnesium (Mg)..................9.87% Water Soluble Magnesium (Mg)
Sulfer(S).................................................12.98%Combined Sulfer(S)
Directions for use (Fertilizer)
Lawns: Apply at a rate of 1/2 pound per one thousand square feet of lawn.
Garden Plants & Shrubbery: 2 tablespoons around the plant base every 2-4 weeks.
Trees: 2 tablespoons over the root zone every 4 months.
Tomatoes: 1 teaspoon per foot plant height every 2 weeks.
Evergreen, Azaleas & Rhododendrons: 1 tablespoon over root zone every 2-4 weeks.
Garden Startup: Mix 1 cup per 100 sq. ft of soil prior to planting.
Houseplants: 1 teaspoon per gallon of water and feed plants every 1-4 weeks.
An Epsom Salt bath or soak has been trusted as a natural remedy for minor aches and sprains for hundreds of years. Epsom Salt can help relieve tired, achy muscles by providing Magnesium to help reduce swelling, and Sulfate which helps flush toxins from proteins in joints. Epsom Salt can also be consumed as a natural saline laxative for occassional relief.
EPSOM SALT WITHOUT SCENT CAN BE APPLIED FOR BOTH INTERNAL AND EXTERNAL USE, SCENTED EPSOM SALTS CAN BE APPLIED FOR EXTERNAL USE ONLY.
- SPL UNCLASSIFIED SECTION
- INACTIVE INGREDIENT
- Package Label
-
INGREDIENTS AND APPEARANCE
HEALTH SMART NATURAL EPSOM SALT
magnesium sulfate granule, for solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:52862-201 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength MAGNESIUM SULFATE (UNII: DE08037SAB) (MAGNESIUM CATION - UNII:T6V3LHY838) MAGNESIUM SULFATE 1 g in 1 g Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:52862-201-16 454 g in 1 POUCH Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part334 08/01/2013 Labeler - International Wholesale, Inc. (161872676)