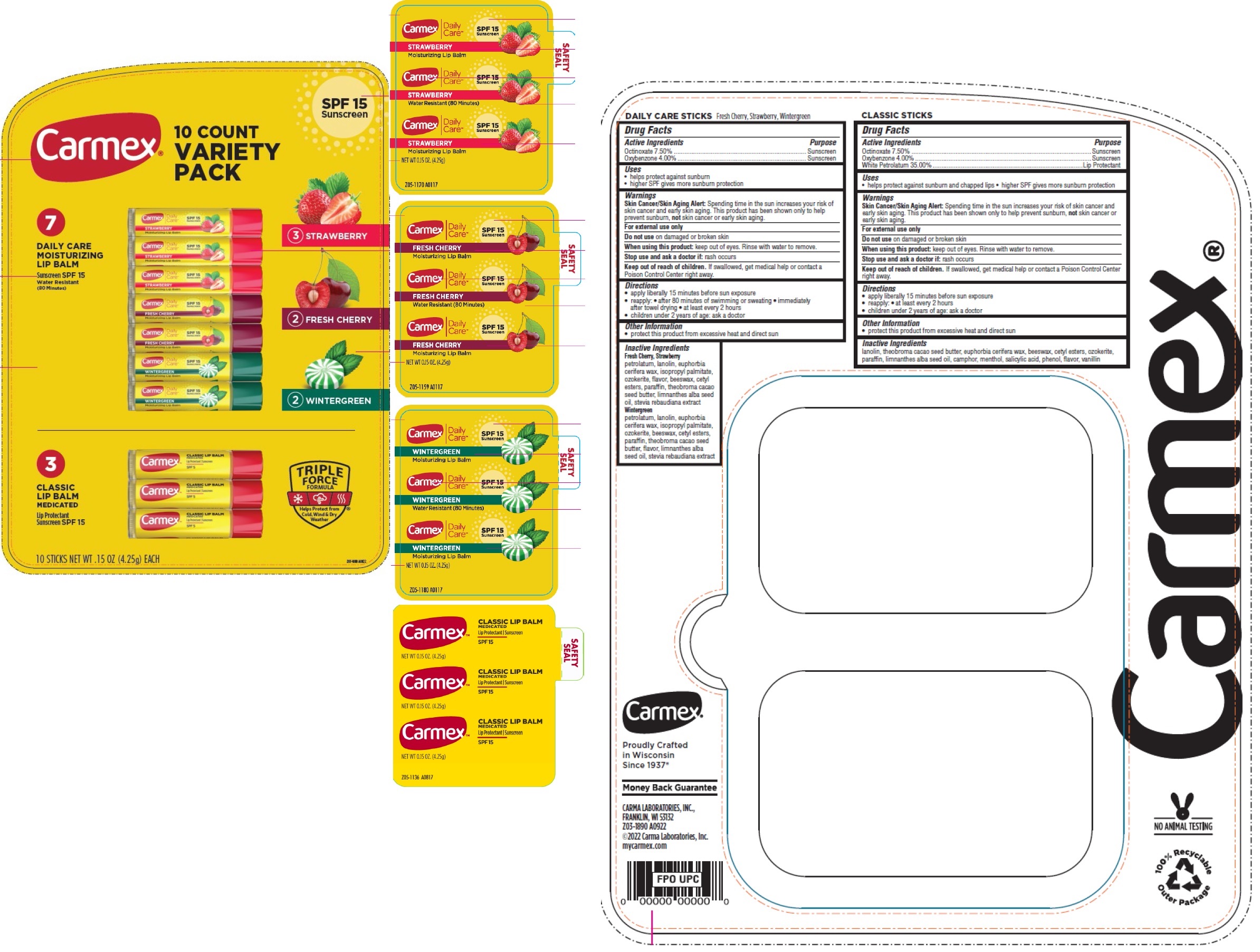
Label: CARMEX 10 COUNT VARIETY PACK SPF 15 SUNSCREEN- octinoxate, oxybenzone, petrolatum kit
-
NDC Code(s):
10210-0023-1,
10210-0028-1,
10210-0031-1,
10210-0033-1, view more10210-0057-1
- Packager: Carma Laboratories, Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 25, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active Ingredients
- Uses
- Warnings
- Directions
- Other Information
-
Inactive Ingredients
Fresh Cherry, Strawberry
petrolatum, lanolin, euphorbia cerifera wax, isopropyl palmitate, ozokerite, flavor, beeswax, cetyl esters, paraffin, theobroma cacao seed butter, limnanthes alba seed oil, stevia rebaudiana extract
Wintergreen
petrolatum, lanolin, euphorbia cerifera wax, isopropyl palmitate, ozokerite, beeswax, cetyl esters, paraffin, theobroma cacao seed butter, flavor, limnanthes alba seed oil, stevia rebaudiana extract
- Drug Facts
- Active Ingredients
- Uses
- Warnings
- Directions
- Other Information
- Inactive Ingredients
- Package Labeling:
-
INGREDIENTS AND APPEARANCE
CARMEX 10 COUNT VARIETY PACK SPF 15 SUNSCREEN
octinoxate, oxybenzone, petrolatum kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:10210-0057 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0057-1 1 in 1 TRAY 01/01/2023 1 1 in 1 KIT Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 3 CYLINDER 12.75 g Part 2 2 CYLINDER 8.5 g Part 3 2 CYLINDER 8.5 g Part 4 3 CYLINDER 12.75 g Part 1 of 4 CARMEX DAILY CARE MOISTURIZING LIP BALM STRAWBERRY SPF 15
octinoxate, oxybenzone salveProduct Information Item Code (Source) NDC:10210-0031 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) LANOLIN (UNII: 7EV65EAW6H) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) YELLOW WAX (UNII: 2ZA36H0S2V) CETYL ESTERS WAX (UNII: D072FFP9GU) PARAFFIN (UNII: I9O0E3H2ZE) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0031-1 4.25 g in 1 CYLINDER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2023 Part 2 of 4 CARMEX DAILY CARE MOISTURIZING LIP BALM FRESH CHERRY SPF 15
octinoxate, oxybenzone salveProduct Information Item Code (Source) NDC:10210-0028 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) LANOLIN (UNII: 7EV65EAW6H) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) YELLOW WAX (UNII: 2ZA36H0S2V) CETYL ESTERS WAX (UNII: D072FFP9GU) PARAFFIN (UNII: I9O0E3H2ZE) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0028-1 4.25 g in 1 CYLINDER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2023 Part 3 of 4 CARMEX DAILY CARE MOISTURIZING LIP BALM WINTERGREEN SPF 15
octinoxate, oxybenzone salveProduct Information Item Code (Source) NDC:10210-0033 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 75 mg in 1 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 40 mg in 1 g Inactive Ingredients Ingredient Name Strength PETROLATUM (UNII: 4T6H12BN9U) LANOLIN (UNII: 7EV65EAW6H) ISOPROPYL PALMITATE (UNII: 8CRQ2TH63M) YELLOW WAX (UNII: 2ZA36H0S2V) CETYL ESTERS WAX (UNII: D072FFP9GU) PARAFFIN (UNII: I9O0E3H2ZE) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0033-1 4.25 g in 1 CYLINDER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2023 Part 4 of 4 CARMEX CLASSIC LIP BALM MEDICATED SPF 15
octinoxate, oxybenzone, petrolatum salveProduct Information Item Code (Source) NDC:10210-0023 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 7.5 g in 100 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 4 g in 100 g WHITE PETROLATUM (UNII: B6E5W8RQJ4) (WHITE PETROLATUM - UNII:B6E5W8RQJ4) WHITE PETROLATUM 35 g in 100 g Inactive Ingredients Ingredient Name Strength LANOLIN (UNII: 7EV65EAW6H) YELLOW WAX (UNII: 2ZA36H0S2V) CETYL ESTERS WAX (UNII: D072FFP9GU) PARAFFIN (UNII: I9O0E3H2ZE) MEADOWFOAM SEED OIL (UNII: 412ZHA4T4Y) CAMPHOR (SYNTHETIC) (UNII: 5TJD82A1ET) MENTHOL, UNSPECIFIED FORM (UNII: L7T10EIP3A) SALICYLIC ACID (UNII: O414PZ4LPZ) PHENOL (UNII: 339NCG44TV) VANILLIN (UNII: CHI530446X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:10210-0023-1 4.25 g in 1 CYLINDER; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2023 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M020 01/01/2023 Labeler - Carma Laboratories, Inc. (006090153)