Label: KAOLIN PECTIN 4- kaolin pectin suspension
- NDC Code(s): 13985-526-01
- Packager: MWI/VetOne
- Category: OTC ANIMAL DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated September 16, 2013
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
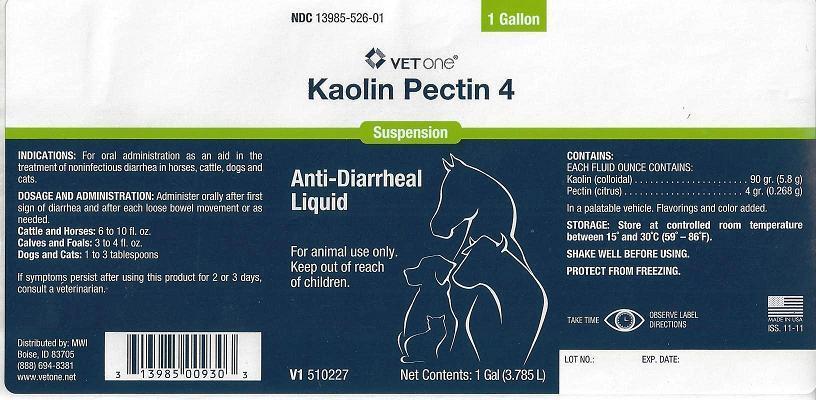
- NDC 13985-526-01 1 GallonVET ONEKaolin Pectin 4SuspensionAnti-Diarrheal Liquid
- INDICATIONS:
-
DOSAGE AND ADMINISTRATION:
Administer orally after first sign of diarrhea and after each loose bowel movement or as needed.
Cattle and Horses: 6 to 10 fl. oz.
Calves and Foals: 3 to 4 fl. oz.
Dogs and Cats: 1 to 3 tablespoons
If symptoms persist after using this product for 2 or 3 days, consult a veterinarian.
Distributed by: MWI
Boise, ID 83705
(888) 694-8381
- CONTAINS:
- STORAGE:
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
KAOLIN PECTIN 4
kaolin pectin suspensionProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:13985-526 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength KAOLIN (UNII: 24H4NWX5CO) (KAOLIN - UNII:24H4NWX5CO) KAOLIN 196 mg in 1 mL PECTIN, CITRUS (UNII: 47EQO8LE7H) (PECTIN - UNII:89NA02M4RX) PECTIN, CITRUS 9 mg in 1 mL Inactive Ingredients Ingredient Name Strength GUAR GUM (UNII: E89I1637KE) METHYLPARABEN (UNII: A2I8C7HI9T) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SACCHARIN SODIUM (UNII: SB8ZUX40TY) PROPYLPARABEN (UNII: Z8IX2SC1OH) FD&C RED NO. 40 (UNII: WZB9127XOA) WATER (UNII: 059QF0KO0R) Product Characteristics Color Score Shape Size Flavor STRAWBERRY Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:13985-526-01 3785 mL in 1 BOTTLE Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug other 01/06/2012 Labeler - MWI/VetOne (019926120)