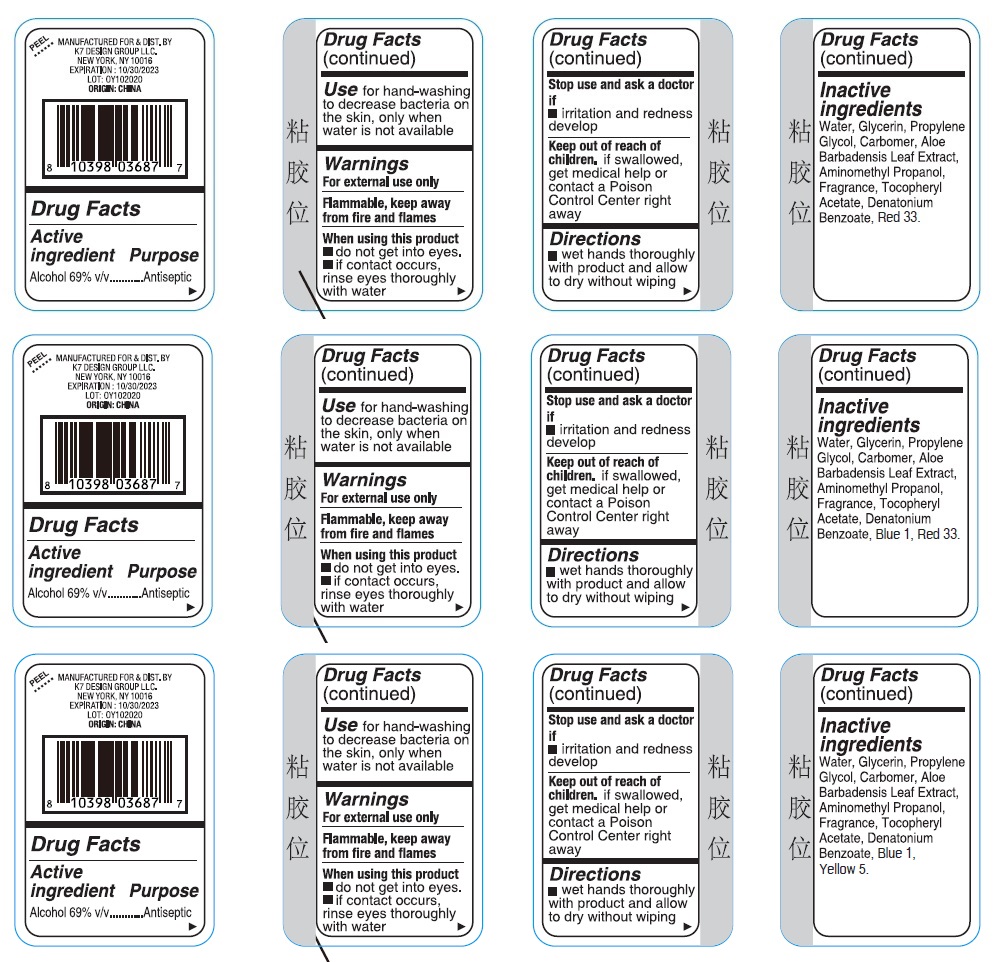
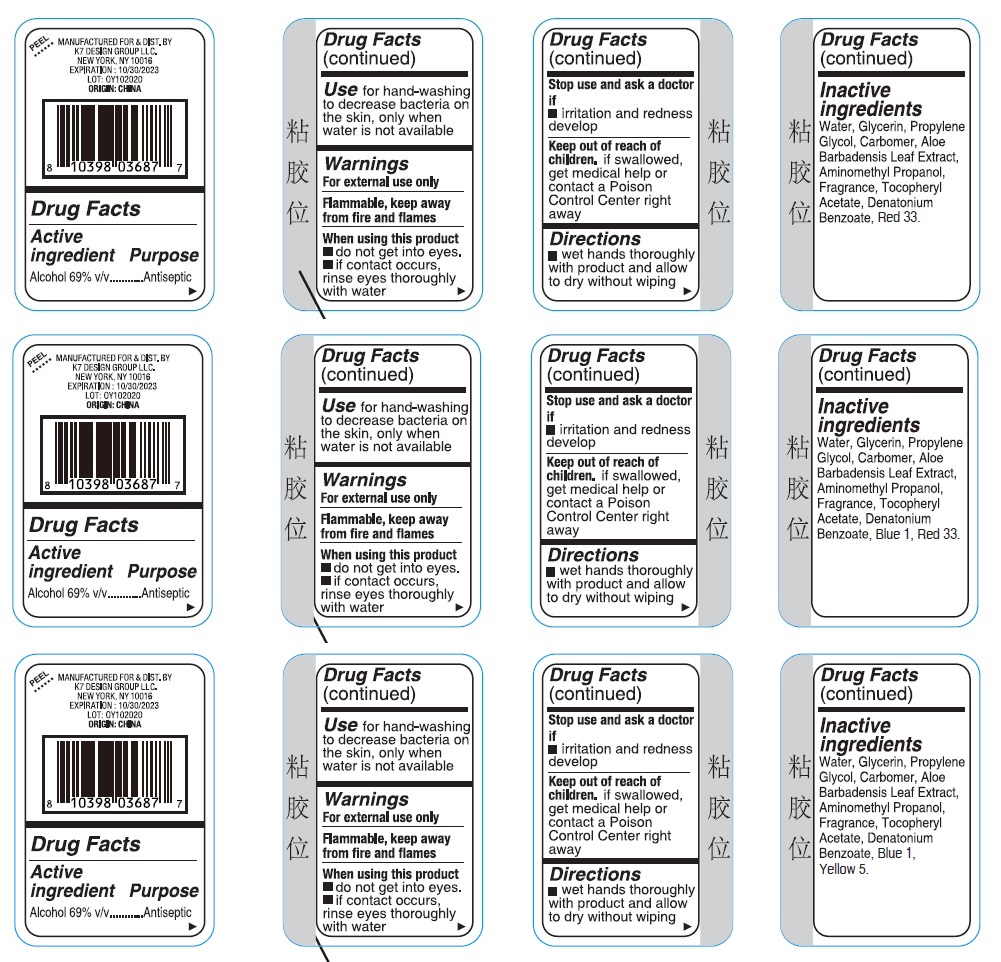
Label: ULTRA DEFENSE SANI SMART 3PK HAND SANITIZER- alcohol kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 74177-851-01 - Packager: K7 Design Group Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph not final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 13, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Active ingredient
- Purpose
- Use
- Warnings
- Directions
-
Inactive ingredients
Strawberry
Water, Glycerin, Propylene Glycol, Carbomer, Aloe Barbadensis Leaf Extract, Aminomethyl Propanol, Fragrance, Tocopheryl Acetate, Denatonium Benzoate, Red 33.
Raspberry
Water, Glycerin, Propylene Glycol, Carbomer, Aloe Barbadensis Leaf Extract, Aminomethyl Propanol, Fragrance, Tocopheryl Acetate, Denatonium Benzoate, Blue 1, Red 33.
Kiwi Berry
Water, Glycerin, Propylene Glycol, Carbomer, Aloe Barbadensis Leaf Extract, Aminomethyl Propanol, Fragrance, Tocopheryl Acetate, Denatonium Benzoate, Blue 1, Yellow 5.
- Company Information
- Product Packaging
-
INGREDIENTS AND APPEARANCE
ULTRA DEFENSE SANI SMART 3PK HAND SANITIZER
alcohol kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:74177-851 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:74177-851-01 1 in 1 KIT; Type 0: Not a Combination Product 10/13/2020 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BOTTLE, PLASTIC 59 mL Part 2 1 BOTTLE, PLASTIC 59 mL Part 3 1 BOTTLE, PLASTIC 59 mL Part 1 of 3 ULTRA DEFENSE SANI SMART KIWI BERRY HAND SANITIZER
alcohol gelProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 69 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) GLYCERIN (UNII: PDC6A3C0OX) ALOE VERA LEAF (UNII: ZY81Z83H0X) DENATONIUM BENZOATE (UNII: 4YK5Z54AT2) FD&C YELLOW NO. 5 (UNII: I753WB2F1M) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) AMINOMETHYLPROPANOL (UNII: LU49E6626Q) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 59 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 10/13/2020 Part 2 of 3 ULTRA DEFENSE SANI SMART STRAWBERRY HAND SANITIZER
alcohol gelProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 69 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) GLYCERIN (UNII: PDC6A3C0OX) ALOE VERA LEAF (UNII: ZY81Z83H0X) DENATONIUM BENZOATE (UNII: 4YK5Z54AT2) D&C RED NO. 33 (UNII: 9DBA0SBB0L) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) AMINOMETHYLPROPANOL (UNII: LU49E6626Q) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 59 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 10/13/2020 Part 3 of 3 ULTRA DEFENSE SANI SMART RASPBERRY HAND SANITIZER
alcohol gelProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 69 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) GLYCERIN (UNII: PDC6A3C0OX) ALOE VERA LEAF (UNII: ZY81Z83H0X) DENATONIUM BENZOATE (UNII: 4YK5Z54AT2) D&C RED NO. 33 (UNII: 9DBA0SBB0L) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) AMINOMETHYLPROPANOL (UNII: LU49E6626Q) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 59 mL in 1 BOTTLE, PLASTIC; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 10/13/2020 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 10/13/2020 Labeler - K7 Design Group Inc. (080357784)