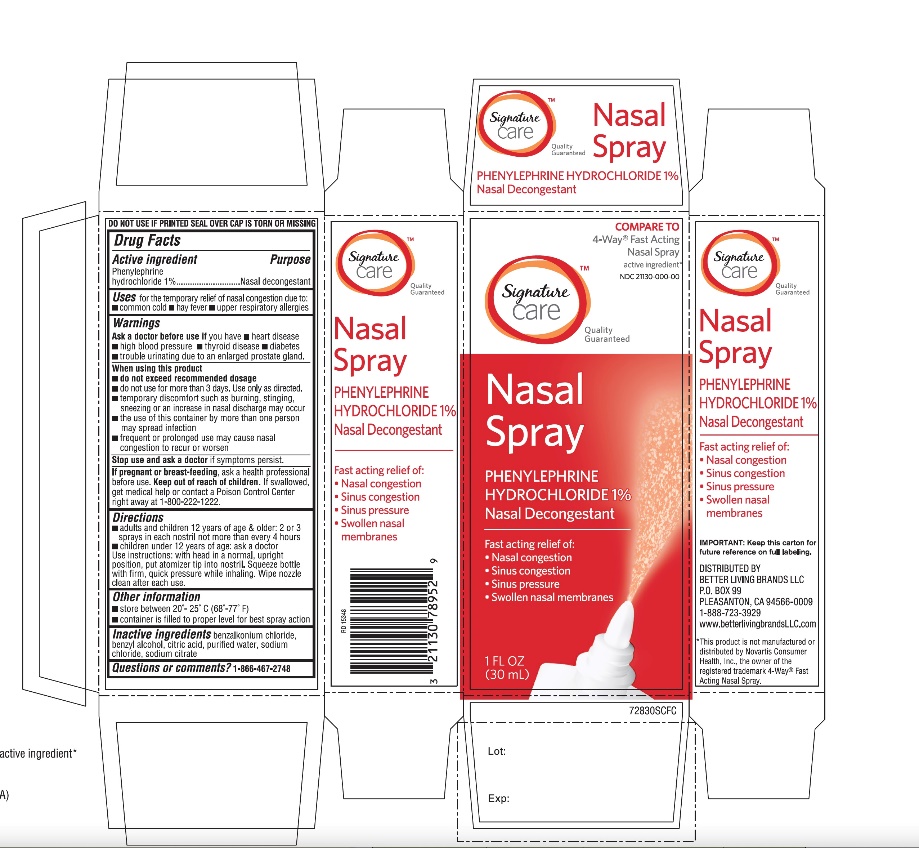
Label: BETTER LIVING- phenylephrine hydrochloride spray
- NDC Code(s): 21130-728-30
- Packager: BETTER LIVING BRANDS LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated November 14, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active ingredient
- Purpose
- Uses
-
Warnings
Ask a doctor before use if you have
- ▪
- heart disease
- ▪
- high blood pressure
- ▪
- thyroid disease
- ▪
- diabetes
- ▪
- trouble urinating due to an enlarged prostate gland
When using this product
- ▪
- do not exceed recommended dosage
- ▪
- do not use for more than 3 days.
- ▪
- use only as directed.
- ▪
- frequent or prolonged use may cause nasal congestion to recur or worsen
- ▪
- temporary discomfort such as burning, stinging, sneezing or an increase in nasal discharge may occur
- ▪
- the use of this container by more than one person may spread infection
-
Directions
- ▪
- adults and children 12 years of age & over: 2 or 3 sprays in each nostril not more often than every 4 hours
- ▪
- children under 12 years of age: ask a doctor
- ▪
- Use instructions: with head in a normal, upright position, put atomizer tip into nostril. Squeeze bottle with firm, quick pressure while inhaling. Wipe nozzle clean after each use.
- Other information
- Inactive ingredients
- Questions or comments?
-
Principal Display Panel
Compare to the active ingredient in 4-Way Fast Acting Nasal Spray *
NDC 21130-728-30
Signature Care™
Quality Guaranteed
Nasal Spray
Nasal Decongestant
Phenylephrine Hydrochloride
Fast acting relief of:
- •
- Nasal congestion
- •
- Sinus congestion
- •
- Sinus pressure
- •
- Swollen nasal membranes
1 FL OZ (30mL)
DO NOT USE IF PRINTED SEAL OVER CAP IS TORN OR MISSING
Distributed By
IMPORTANT: Keep this carton for future reference on full labeling.
*This product is not manufactured or distributed by GSK Consumer Healthcare, the distributor of 4-Way® Fast acting Nasal Spray.
-
INGREDIENTS AND APPEARANCE
BETTER LIVING
phenylephrine hydrochloride sprayProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:21130-728 Route of Administration NASAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 1 g in 100 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) BENZYL ALCOHOL (UNII: LKG8494WBH) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) WATER (UNII: 059QF0KO0R) SODIUM CHLORIDE (UNII: 451W47IQ8X) SODIUM CITRATE, UNSPECIFIED FORM (UNII: 1Q73Q2JULR) Product Characteristics Color YELLOW ((Clear to slightly yellow)) Score Shape Size Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:21130-728-30 1 in 1 CARTON 04/17/2019 1 30 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 04/17/2019 Labeler - BETTER LIVING BRANDS LLC (009137209)