Label: CETIRIZINE HYDROCHLORIDE tablet
- NDC Code(s): 56062-939-30, 56062-939-54, 56062-939-60
- Packager: Publix Super Markets Inc
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: Abbreviated New Drug Application
Drug Label Information
Updated August 19, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENT (IN EACH TABLET)
- PURPOSE
- USES
-
WARNINGS
Do not use
If you have ever had an allergic reaction to this product or any of its ingredients or to an antihistamine containing hydroxyzine.
Ask a doctor before use if you have
Liver or kidney disease. Your doctor should determine if you need a different dose.
When using this product
- •
- drowsiness may occur
- •
- avoid alcoholic drinks
- •
- alcohol, sedatives, and tranquilizers may increase drowsiness
- •
- be careful when driving a motor vehicle or operating machinery
Stop use and ask a doctor if
An allergic reaction to this product occurs. Seek medical help right away.
-
DIRECTIONS
adults and children 6 years and over
one 10 mg tablet once daily; do not take more than one 10 mg tablet in 24 hours. A 5 mg product may be appropriate for less severe symptoms.
adults 65 years and over
ask a doctor
children under 6 years of age
ask a doctor
consumers with liver or kidney disease
ask a doctor
- Other information
- INACTIVE INGREDIENTS
-
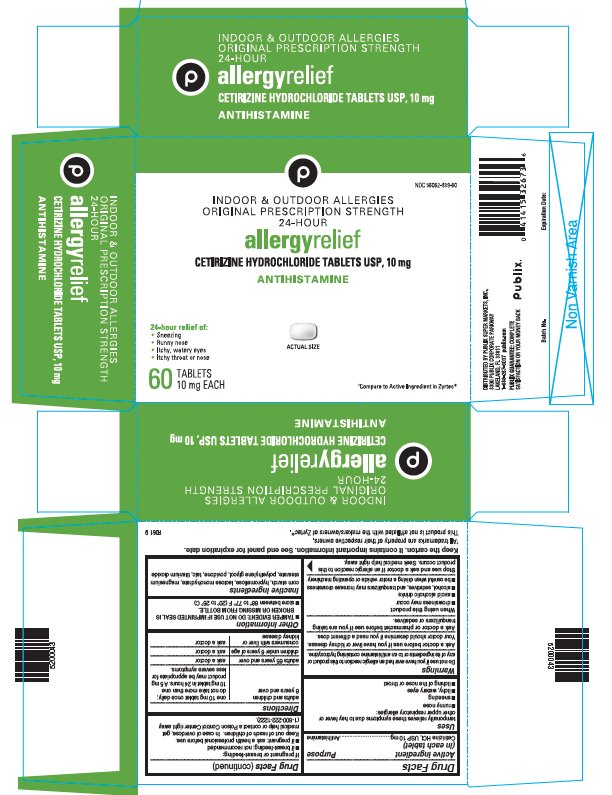
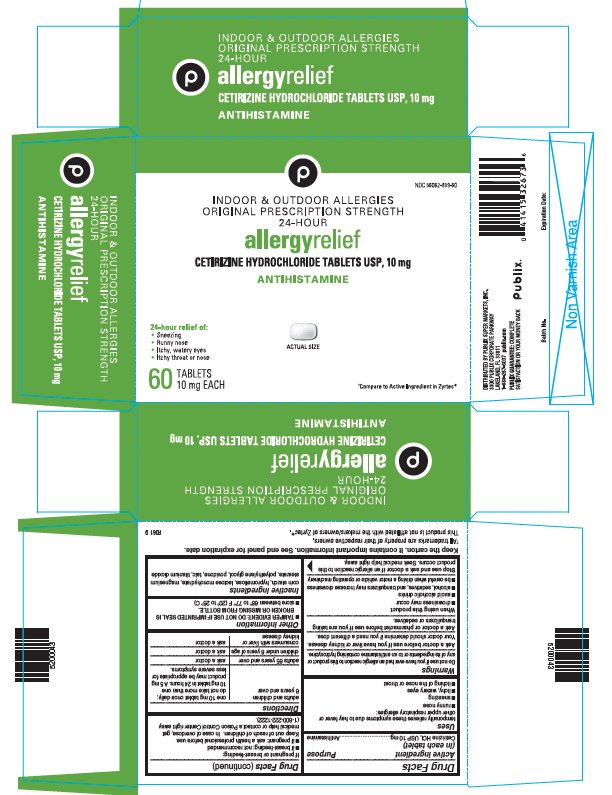
PRINCIPAL DISPLAY PANEL - 10 mg Tablet Bottle Carton
NDC 56062-939-60
INDOOR & OUTDOOR ALLERGIES
ORIGINAL PRESCRIPTION STRENGTH
24-HOURallergyrelief
CETIRIZINE HYDROCHLORIDE TABLETS USP, 10 mg
ANTIHISTAMINE
24-hour relief of:
- •
- Sneezing
- •
- Runny nose
- •
- Itchy, watery eyes
- •
- Itchy throat or nose
ACTUAL SIZE
60
TABLETS
10 mg EACH†Compare to Active Ingredient in Zyrtec®
-
INGREDIENTS AND APPEARANCE
CETIRIZINE HYDROCHLORIDE
cetirizine hydrochloride tabletProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:56062-939 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CETIRIZINE HYDROCHLORIDE (UNII: 64O047KTOA) (CETIRIZINE - UNII:YO7261ME24) CETIRIZINE HYDROCHLORIDE 10 mg Inactive Ingredients Ingredient Name Strength STARCH, CORN (UNII: O8232NY3SJ) HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) LACTOSE MONOHYDRATE (UNII: EWQ57Q8I5X) MAGNESIUM STEARATE (UNII: 70097M6I30) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) POVIDONE, UNSPECIFIED (UNII: FZ989GH94E) TALC (UNII: 7SEV7J4R1U) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color white Score no score Shape RECTANGLE (Rounded-Off) Size 9mm Flavor Imprint Code R152 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:56062-939-54 14 in 1 BLISTER PACK; Type 0: Not a Combination Product 12/27/2007 2 NDC:56062-939-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 12/27/2007 3 NDC:56062-939-60 60 in 1 BOTTLE; Type 0: Not a Combination Product 12/27/2007 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA077498 12/27/2007 Labeler - Publix Super Markets Inc (006922009) Registrant - Sun Pharmaceutical Industries Inc. (146974886) Establishment Name Address ID/FEI Business Operations Ohm Laboratories Inc. 184769029 manufacture(56062-939)