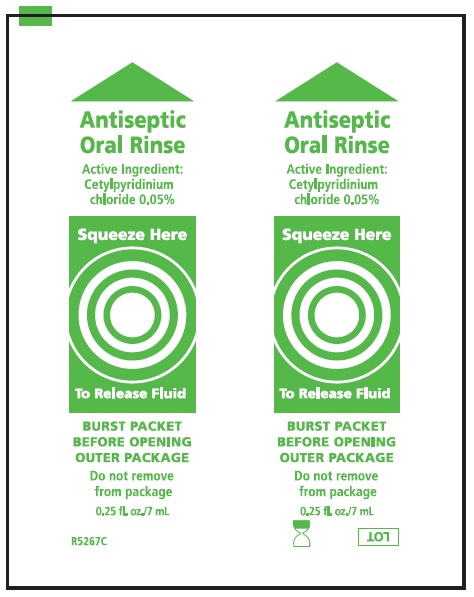
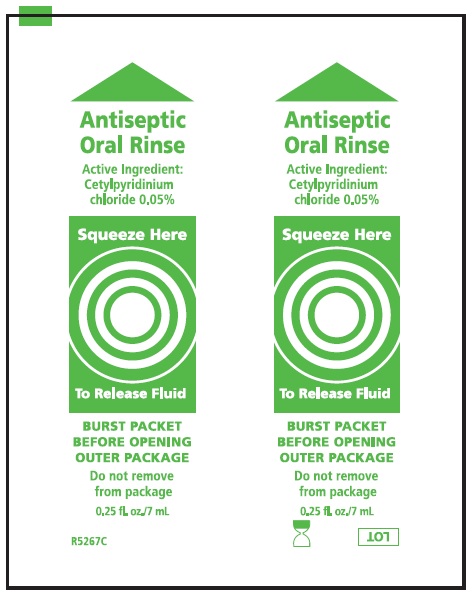
Label: SUCTION TOOTHBRUSHES- cetylpyridinium chloride kit
-
Contains inactivated NDC Code(s)
NDC Code(s): 53462-175-60, 53462-273-60 - Packager: Sage Products LLC
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated April 26, 2018
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Drug Facts
- Uses
- INDICATIONS & USAGE
- Warnings
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
-
Directions
- Before opening, turn package over, burst solution packet with thumbs.
- Peel lid to open.
- Remove toothbrush and attach to suction line.
- Clean teeth and oral cavity for approximately one minute.
- To suction, place thumb over port.
- Repeat cleaning with other toothbrush as needed.
- Discard after use.
- Apply Mouth Moisturizer as needed.
- Use up to 4 times daily, or as directed by a dentist or doctor.
- Children under 12 years of age should be supervised in the use of this product.
- Children under 3 years of age, consult a dentist or doctor.
- Use a bite block when performing oral care on patients with altered levels of consciousness or those who cannot comprehend commands.
- Ensure foam is intact after use. If not, remove any particles from oral cavity.
- Inactive Ingredients
- Questions
- SPL UNCLASSIFIED SECTION
- Suction Toothbrushes
- Antispetic Oral Rinse
-
INGREDIENTS AND APPEARANCE
SUCTION TOOTHBRUSHES
cetylpyridinium chloride kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:53462-273 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:53462-273-60 1 in 1 KIT; Type 1: Convenience Kit of Co-Package 06/30/2006 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 PACKET 7 mL Part 2 1 PACKET 2 g Part 3 1 Part 1 of 3 ANTISEPTIC ORAL RINSE
cetylpyridinium chloride mouthwashProduct Information Item Code (Source) NDC:53462-175 Route of Administration BUCCAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CETYLPYRIDINIUM CHLORIDE (UNII: D9OM4SK49P) (CETYLPYRIDINIUM - UNII:CUB7JI0JV3) CETYLPYRIDINIUM CHLORIDE 0.5 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) PEPPERMINT (UNII: V95R5KMY2B) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) POLYSORBATE 80 (UNII: 6OZP39ZG8H) POLYSORBATE 20 (UNII: 7T1F30V5YH) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 KIT 1 NDC:53462-175-60 7 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 06/21/2000 Part 2 of 3 MOUTH MOISTURIZER
other oral hygiene productsProduct Information Other Ingredients Ingredient Kind Ingredient Name Quantity INGR COCONUT OIL (UNII: Q9L0O73W7L) INGR XYLITOL (UNII: VCQ006KQ1E) INGR CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) INGR .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) INGR WATER (UNII: 059QF0KO0R) INGR POLYSORBATE 20 (UNII: 7T1F30V5YH) INGR POLYSORBATE 80 (UNII: 6OZP39ZG8H) INGR POTASSIUM SORBATE (UNII: 1VPU26JZZ4) INGR CETYLPYRIDINIUM CHLORIDE (UNII: D9OM4SK49P) INGR CARBOMER HOMOPOLYMER TYPE B (ALLYL PENTAERYTHRITOL CROSSLINKED) (UNII: HHT01ZNK31) INGR SPEARMINT OIL (UNII: C3M81465G5) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 1 in 1 KIT 1 2 g in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Cosmetic Part 3 of 3 SODIUM BICARBONATE
other oral hygiene productsProduct Information Other Ingredients Ingredient Kind Ingredient Name Quantity INGR SODIUM BICARBONATE (UNII: 8MDF5V39QO) INGR CARBOXYMETHYLCELLULOSE SODIUM (UNII: K679OBS311) INGR SODIUM LAURYL SULFATE (UNII: 368GB5141J) INGR SODIUM BENZOATE (UNII: OJ245FE5EU) INGR WATER (UNII: 059QF0KO0R) INGR SACCHARIN SODIUM (UNII: SB8ZUX40TY) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Cosmetic Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part356 06/30/2006 Labeler - Sage Products LLC (054326178) Registrant - Sage Products LLC (054326178)