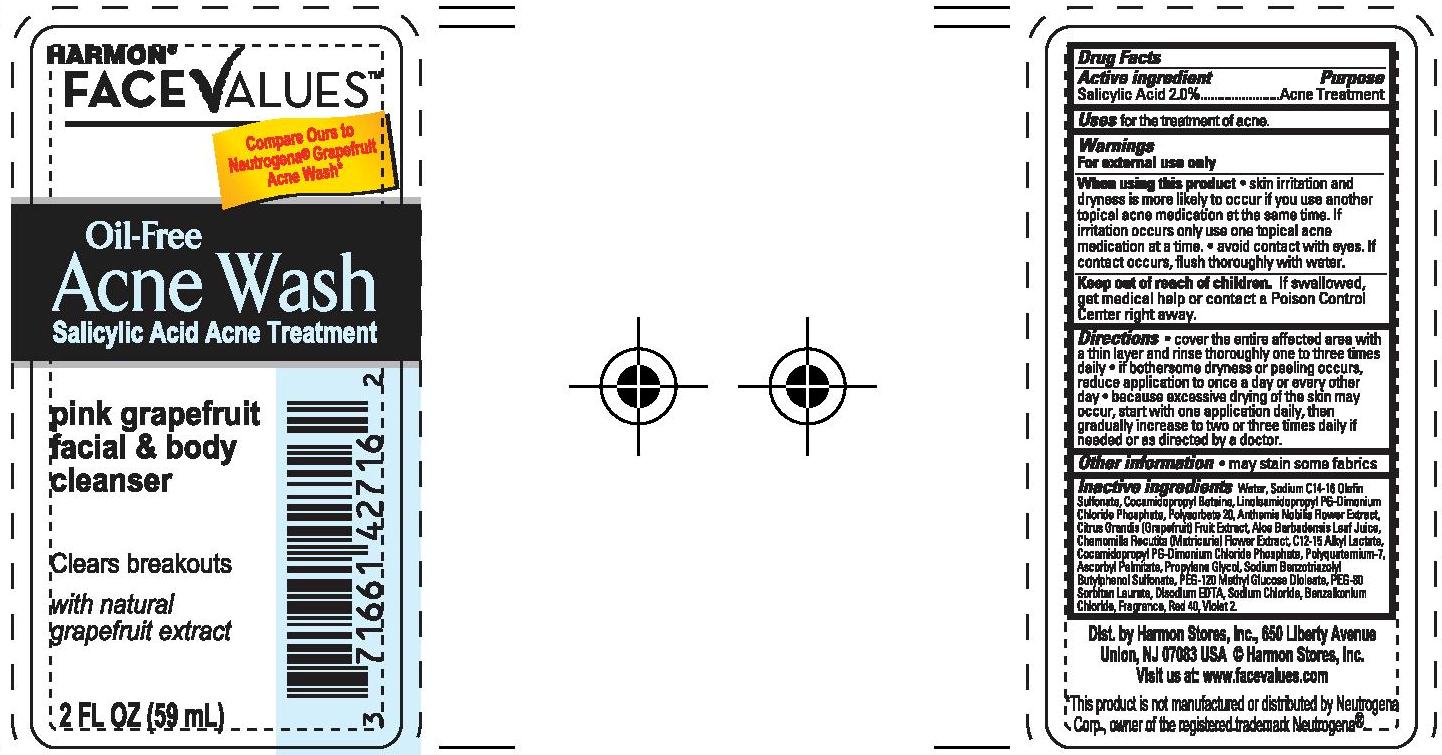
Label: ACNE WASH- salicylic acid liquid
- NDC Code(s): 63940-053-54, 63940-053-61
- Packager: Harmon Stores Inc.
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated March 19, 2020
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredients
- Purpose
- Uses
- Warnings
- WHEN USING
- KEEP OUT OF REACH OF CHILDREN
-
Directions
- cover the entire affected area with a thin layer and rinse thoroughly one to three times daily
- because excessive drying of the skin may occur, start with one application daily, then gradually increase to two or three times daily if needed or as directed by a doctor
- if bothersome dryness or peeling occurs, reduce application to once a day or every other day
- Other information
-
Inactive Ingredients
water, sodium C14-16 olefin sulfonate, cocamidopropyl betaine, sodium chloride, linoleamidopropyl PG-dimonium chloride phosphate, polysorbate 20, anthemis nobilis flower extract, citrus grandis (grapefruit) fruit extract, aloe barbadensis leaf juice, chamomilla recutita (matricaria) flower extract, C12-15 alkyl lactate, cocamidopropyl PG-dimononium chloride phosphate, polyquaternium-7, ascorbyl palmitate, propylene glycol, sodium benzotriazolyl butylphenol sulfonate, PEG-120 methyl glucose dioleate, PEG-80 sorbitan laurate, disodium EDTA, benzalkonium chloride, fragrance, red 40, Ext. Violet 2
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ACNE WASH
salicylic acid liquidProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:63940-053 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength SALICYLIC ACID (UNII: O414PZ4LPZ) (SALICYLIC ACID - UNII:O414PZ4LPZ) SALICYLIC ACID 20 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) SODIUM C14-16 OLEFIN SULFONATE (UNII: O9W3D3YF5U) COCAMIDOPROPYL BETAINE (UNII: 5OCF3O11KX) SODIUM CHLORIDE (UNII: 451W47IQ8X) LINOLEAMIDOPROPYL PG-DIMONIUM CHLORIDE PHOSPHATE (UNII: 5Q87K461JO) POLYSORBATE 20 (UNII: 7T1F30V5YH) CHAMAEMELUM NOBILE FLOWER (UNII: O2T154T6OG) PUMMELO (UNII: ET1TN5W71X) ALOE VERA LEAF (UNII: ZY81Z83H0X) CHAMOMILE (UNII: FGL3685T2X) C12-15 ALKYL LACTATE (UNII: GC844VRD7E) COCAMIDOPROPYL PG-DIMONIUM CHLORIDE PHOSPHATE (UNII: H2KVQ74JM4) POLYQUATERNIUM-7 (70/30 ACRYLAMIDE/DADMAC; 1600 KD) (UNII: 0L414VCS5Y) ASCORBYL PALMITATE (UNII: QN83US2B0N) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM BENZOTRIAZOLYL BUTYLPHENOL SULFONATE (UNII: 0LA2QC9O3Z) PEG-120 METHYL GLUCOSE DIOLEATE (UNII: YM0K64F20V) PEG-80 SORBITAN LAURATE (UNII: 239B50Y732) EDETATE DISODIUM (UNII: 7FLD91C86K) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) FD&C RED NO. 40 (UNII: WZB9127XOA) EXT. D&C VIOLET NO. 2 (UNII: G5UX3K0728) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63940-053-54 269 mL in 1 BOTTLE, PUMP; Type 0: Not a Combination Product 02/05/2016 2 NDC:63940-053-61 59 mL in 1 BOTTLE; Type 0: Not a Combination Product 02/05/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part358H 02/05/2016 Labeler - Harmon Stores Inc. (804085293) Registrant - Fruit of the Earth, Inc. (079559467) Establishment Name Address ID/FEI Business Operations Fruit of the Earth, Inc. 008193513 manufacture(63940-053)