Label: LOTEMAX- loteprednol etabonate gel
- NDC Code(s): 24208-503-07, 24208-503-48, 24208-503-96
- Packager: Bausch & Lomb Incorporated
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated February 28, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use LOTEMAX safely and effectively. See full prescribing information for LOTEMAX.
LOTEMAX® (loteprednol etabonate ophthalmic gel), for topical ophthalmic use
Initial U.S. Approval: 1998INDICATIONS AND USAGE
LOTEMAX is a corticosteroid indicated for the treatment of postoperative inflammation and pain following ocular surgery. (1)
DOSAGE AND ADMINISTRATION
DOSAGE FORMS AND STRENGTHS
LOTEMAX is a sterile preserved ophthalmic gel containing 5 mg of loteprednol etabonate per gram of gel. (3)
CONTRAINDICATIONS
LOTEMAX is contraindicated in most viral diseases of the cornea and conjunctiva including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, and varicella, in mycobacterial infection of the eye and fungal diseases of ocular structures. (4)
WARNINGS AND PRECAUTIONS
- •
- Intraocular pressure (IOP) increase - Prolonged use of corticosteroids may result in glaucoma with damage to the optic nerve, defects in visual acuity and fields of vision. If this product is used for 10 days or longer, IOP should be monitored. (5.1)
- •
- Cataracts - Use of corticosteroids may result in posterior subcapsular cataract formation. (5.2)
- •
- Delayed healing – The use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation. In those diseases causing thinning of the cornea or sclera, perforations have been known to occur with the use of topical steroids. The initial prescription and renewal of the medication order should be made by a physician only after examination of the patient with the aid of magnification such as slit lamp biomicroscopy and, where appropriate, fluorescein staining. (5.3)
- •
- Bacterial infections – Prolonged use of corticosteroids may suppress the host response and thus increase the hazard of secondary ocular infections. In acute purulent conditions, steroids may mask infection or enhance existing infection. (5.4)
- •
- Viral infections – Employment of a corticosteroid medication in the treatment of patients with a history of herpes simplex requires great caution. Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye (including herpes simplex). (5.5)
- •
- Fungal infections – Fungal infections of the cornea are particularly prone to develop coincidentally with long-term local steroid application. Fungus invasion must be considered in any persistent corneal ulceration where a steroid has been used or is in use. (5.6)
ADVERSE REACTIONS
The most common adverse drug reactions (2-5%) were anterior chamber inflammation, eye pain, and foreign body sensation. (6)
To report SUSPECTED ADVERSE REACTIONS, contact Bausch & Lomb Incorporated at 1-800-553-5340 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
See 17 for PATIENT COUNSELING INFORMATION.
Revised: 2/2023
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Intraocular Pressure (IOP) Increase
5.2 Cataracts
5.3 Delayed Healing
5.4 Bacterial Infections
5.5 Viral Infections
5.6 Fungal Infections
5.7 Contact Lens Wear
6 ADVERSE REACTIONS
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
- 1 INDICATIONS AND USAGE
- 2 DOSAGE AND ADMINISTRATION
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
-
5 WARNINGS AND PRECAUTIONS
5.1 Intraocular Pressure (IOP) Increase
Prolonged use of corticosteroids, including LOTEMAX, may result in glaucoma with damage to the optic nerve, defects in visual acuity and fields of vision. Steroids should be used with caution in the presence of glaucoma. If this product is used for 10 days or longer, intraocular pressure should be monitored.
5.3 Delayed Healing
The use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation. In those diseases causing thinning of the cornea or sclera, perforations have been known to occur with the use of topical steroids. The initial prescription and renewal of the medication order should be made by a physician only after examination of the patient with the aid of magnification such as slit lamp biomicroscopy and, where appropriate, fluorescein staining.
5.4 Bacterial Infections
Prolonged use of corticosteroids may suppress the host response and thus increase the hazard of secondary ocular infections. In acute purulent conditions of the eye, steroids may mask infection or enhance existing infection.
5.5 Viral Infections
Employment of a corticosteroid medication in the treatment of patients with a history of herpes simplex requires great caution. Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye (including herpes simplex).
5.6 Fungal Infections
Fungal infections of the cornea are particularly prone to develop coincidentally with long-term local steroid application. Fungus invasion must be considered in any persistent corneal ulceration where a steroid has been used or is in use. Fungal cultures should be taken when appropriate.
-
6 ADVERSE REACTIONS
Adverse reactions associated with ophthalmic steroids include elevated intraocular pressure, which may be associated with infrequent optic nerve damage, visual acuity and field defects, posterior subcapsular cataract formation, delayed wound healing and secondary ocular infection from pathogens including herpes simplex, and perforation of the globe where there is thinning of the cornea or sclera.
The most common adverse drug reactions reported in the clinical trials (2-5%) were anterior chamber inflammation, eye pain, and foreign body sensation.
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no adequate and well-controlled studies with loteprednol etabonate in pregnant women.
Loteprednol etabonate produced teratogenicity at clinically relevant doses in the rabbit and rat when administered orally during pregnancy. Loteprednol etabonate produced malformations when administered orally to pregnant rabbits at doses ≥1.2 times the recommended human ophthalmic dose (RHOD) and to pregnant rats at doses ≥ 30 times the RHOD. In pregnant rats receiving oral doses of loteprednol etabonate during the period equivalent to the last trimester of pregnancy through lactation in humans, survival of offspring was reduced at doses ≥3 times the RHOD. Maternal toxicity was observed in rats at doses ≥ 304 times the RHOD, and a maternal no observed adverse effect level (NOAEL) was established at 30 times the RHOD.
The background risk of major birth defects and miscarriage for the indicated population is unknown. However, the background risk in the U.S. general population of major birth defects is 2 to 4%, and of miscarriage is 15 to 20%, of clinically recognized pregnancies.
Data
Animal Data
Embryofetal studies were conducted in pregnant rabbits administered loteprednol etabonate by oral gavage on gestation days 6 to 18, to target the period of organogenesis. Loteprednol etabonate produced fetal malformations at doses ≥ 0.1 mg/kg (1.2 times the recommended human ophthalmic dose (RHOD) based on body surface area, assuming 100% absorption). Spina bifida (including meningocele) was observed at doses ≥ 0.1 mg/kg, and exencephaly and craniofacial malformations were observed at doses ≥ 0.4 mg/kg (4.9 times the RHOD). At 3 mg/kg (36 times the RHOD), loteprednol etabonate was associated with increased incidences of abnormal left common carotid artery, limb flexures, umbilical hernia, scoliosis, and delayed ossification. Abortion and embryofetal lethality (resorption) occurred at doses ≥ 6 mg/kg (73 times the RHOD). A NOAEL for developmental toxicity was not established in this study. The NOAEL for maternal toxicity in rabbits was 3 mg/kg/day.Embryofetal studies were conducted in pregnant rats administered loteprednol etabonate by oral gavage on gestation days 6 to 15, to target the period of organogenesis. Loteprednol etabonate produced fetal malformations, including absent innominate artery at doses ≥ 5 mg/kg (30 times the RHOD); and cleft palate, agnathia, cardiovascular defects, umbilical hernia, decreased fetal body weight and decreased skeletal ossification at doses ≥ 50 mg/kg (304 times the RHOD). Embryofetal lethality (resorption) was observed at 100 mg/kg (608 times the RHOD). The NOAEL for developmental toxicity in rats was 0.5 mg/kg (3 times the RHOD). Loteprednol etabonate was maternally toxic (reduced body weight gain) at doses of ≥ 50 mg/kg/day. The NOAEL for maternal toxicity was 5 mg/kg.
A peri-/postnatal study was conducted in rats administered loteprednol etabonate by oral gavage from gestation day 15 (start of fetal period) to postnatal day 21 (the end of lactation period). At doses ≥ 0.5 mg/kg (3 times the clinical dose), reduced survival was observed in live-born offspring. Doses ≥ 5 mg/kg (30 times the RHOD) caused umbilical hernia/incomplete gastrointestinal tract. Doses ≥ 50 mg/kg (304 times the RHOD) produced maternal toxicity (reduced body weight gain, death), decreased number of live-born offspring, decreased birth weight, and delays in postnatal development. A developmental NOAEL was not established in this study. The NOAEL for maternal toxicity was 5 mg/kg.
8.2 Lactation
There are no data on the presence of loteprednol etabonate in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered, along with the mother’s clinical need for LOTEMAX and any potential adverse effects on the breastfed infant from LOTEMAX.
8.4 Pediatric Use
The safety and effectiveness of LOTEMAX have been established in the pediatric population. Use of LOTEMAX in this population is supported by evidence from adequate and well-controlled trials of LOTEMAX in adults with additional data from a safety and efficacy trial in pediatric patients from birth to 11 years of age [see Clinical Studies (14)].
-
11 DESCRIPTION
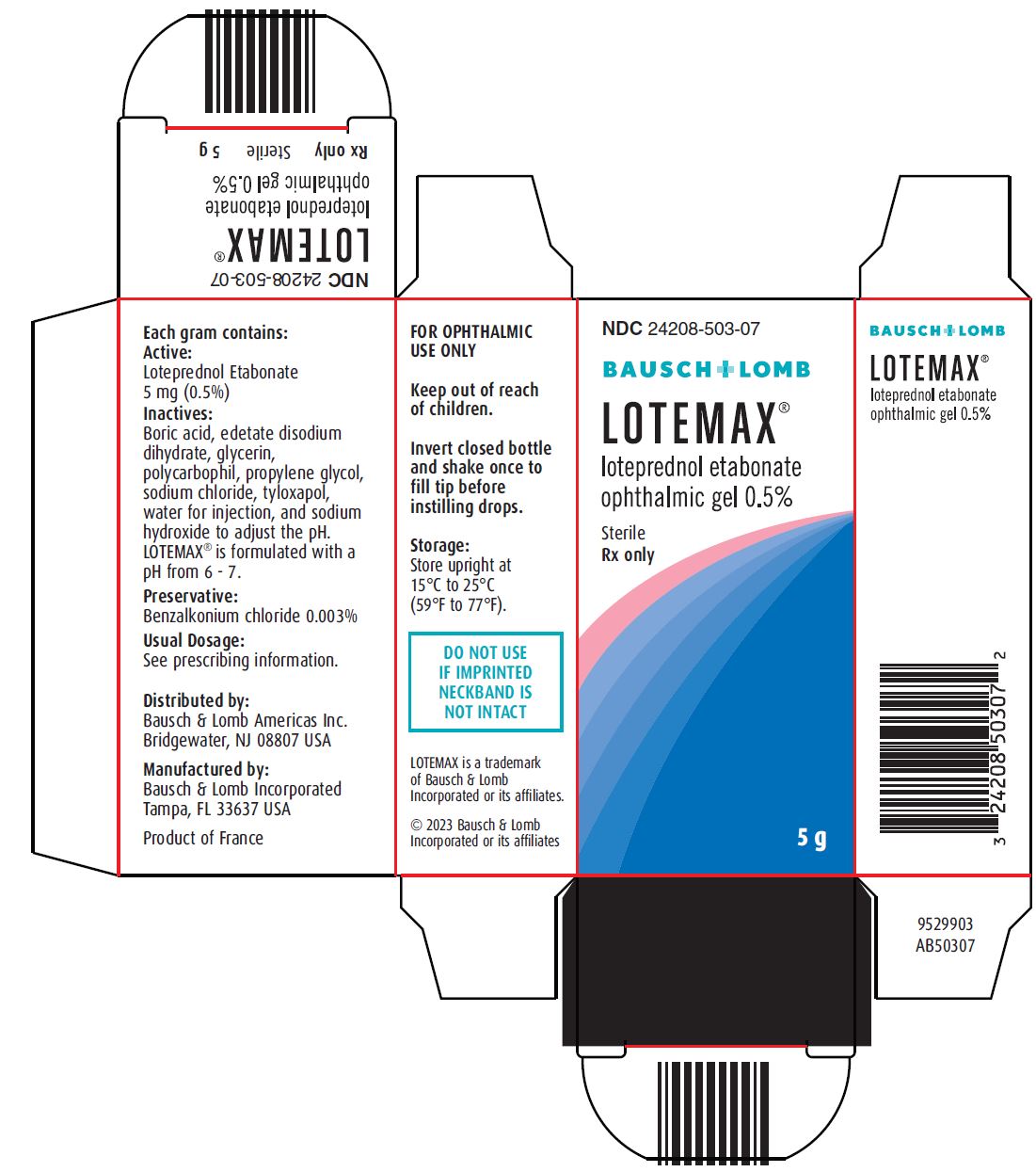
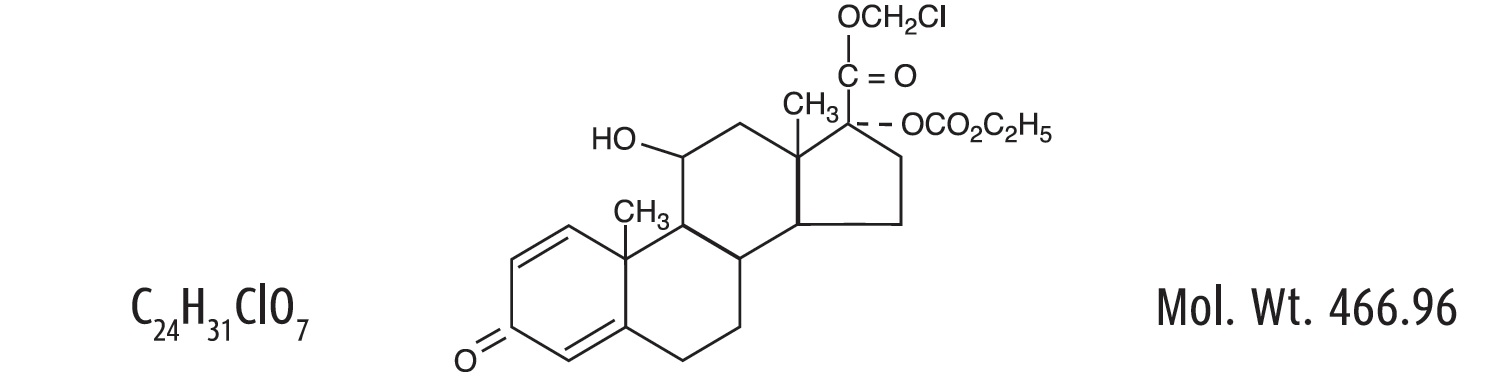
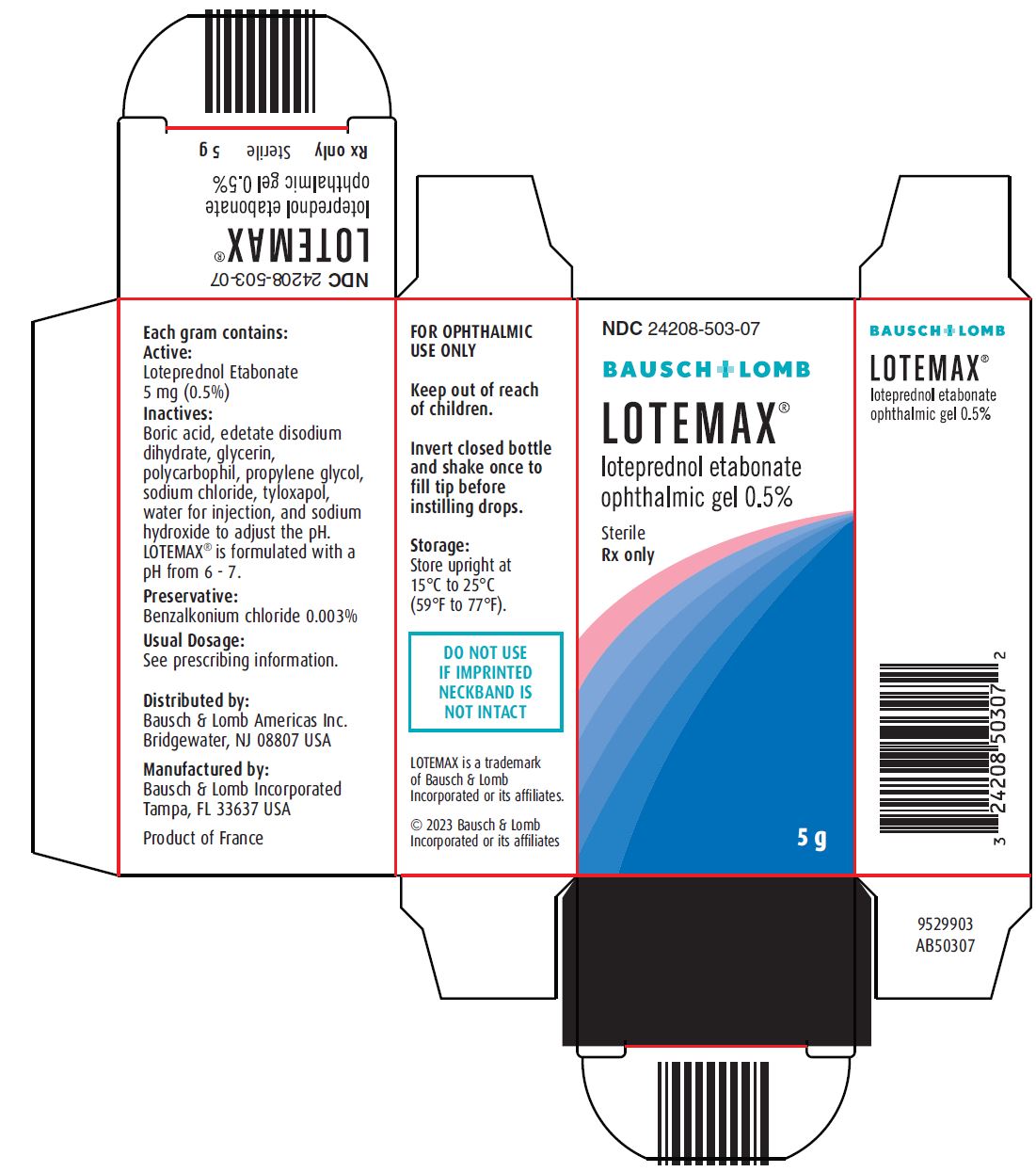
Loteprednol etabonate is a corticosteroid. Its chemical name is chloromethyl 17α-[(ethoxycarbonyl)oxy]-11β-hydroxy-3-oxoandrosta-1,4-diene-17β-carboxylate. Its molecular formula is C24H31ClO7 and its chemical structure is:
LOTEMAX® (loteprednol etabonate ophthalmic gel) 0.5% contains a sterile, topical corticosteroid for ophthalmic use. Loteprednol etabonate is a white to off-white powder.
Each gram contains:
- •
- Active: loteprednol etabonate 5 mg (0.5%)
- •
- Inactives: boric acid, edetate disodium dihydrate, glycerin, polycarbophil, propylene glycol, sodium chloride, tyloxapol, water for injection, and sodium hydroxide to adjust to a pH of between 6 and 7
- •
- Preservative: benzalkonium chloride 0.003%
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Corticosteroids inhibit the inflammatory response to a variety of inciting agents and probably delay or slow healing. They inhibit the edema, fibrin deposition, capillary dilation, leukocyte migration, capillary proliferation, fibroblast proliferation, deposition of collagen, and scar formation associated with inflammation. While glucocorticoids are known to bind to and activate the glucocorticoid receptor, the molecular mechanisms involved in glucocorticoid/glucocorticoid receptor-dependent modulation of inflammation are not clearly established. However, corticosteroids are thought to inhibit prostaglandin production through several independent mechanisms.
12.3 Pharmacokinetics
Loteprednol etabonate is lipid soluble and can penetrate into cells. Loteprednol etabonate is synthesized through structural modifications of prednisolone-related compounds so that it will undergo a predictable transformation to an inactive metabolite. Based upon in vivo and in vitro preclinical metabolism studies, loteprednol etabonate undergoes extensive metabolism to the inactive carboxylic acid metabolites, PJ-91 and PJ-90. The systemic exposure to loteprednol etabonate following ocular administration of LOTEMAX has not been studied in humans.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been conducted to evaluate the carcinogenic potential of loteprednol etabonate. Loteprednol etabonate was not genotoxic in vitro in the Ames test, the mouse lymphoma tk assay, or in a chromosome aberration test in human lymphocytes, or in vivo in the single dose mouse micronucleus assay.
Treatment of female and male rats with doses ≥ 25 mg/kg/day of loteprednol etabonate (152 times the RHOD based on body surface area, assuming 100% absorption) prior to and during mating caused preimplantation loss and decreased the number of live fetuses/live births. The NOAEL for fertility in rats was 5 mg/kg/day (30 times the RHOD).
-
14 CLINICAL STUDIES
Adult Studies
In two randomized, multicenter, double-masked, parallel-group, vehicle-controlled studies in 813 subjects with post-operative inflammation, LOTEMAX was more effective compared to its vehicle in resolving anterior chamber inflammation and pain following cataract surgery. Primary endpoints were complete resolution of anterior chamber cells (cell count of 0) and no pain at post-operative day 8.In these studies, LOTEMAX had a statistically significant higher incidence of subjects with complete clearing of anterior chamber cells (31% vs. 14-16%) and were pain-free at post-operative day 8 (73-76% vs. 42-46%).
Pediatric Study
The safety and effectiveness of LOTEMAX were evaluated in a pediatric study of patients from birth to less than 11 years of age (mean age of 3 years) undergoing cataract surgery. Patients were randomized to receive either LOTEMAX (54 patients) or prednisolone acetate ophthalmic suspension 1% (53 patients) four times daily for 14 days. At Day 14, the percentages of patients with complete clearing of anterior chamber inflammation were 57% in the LOTEMAX group and 63% in the prednisolone group. -
16 HOW SUPPLIED/STORAGE AND HANDLING
LOTEMAX® (loteprednol etabonate ophthalmic gel) 0.5% is a sterile ophthalmic gel supplied in a white low density polyethylene plastic bottle with a white controlled drop tip and a pink polypropylene cap in the following size:
- NDC 24208-503-07 5 g in a 10 mL bottle
Storage: Store upright at 15ºC to 25ºC (59ºF to 77ºF).
-
17 PATIENT COUNSELING INFORMATION
Administration
Invert closed bottle and shake once to fill tip before instilling drops.
Risk of Contamination
Advise patients not to allow the dropper tip to touch any surface, as this may contaminate the gel.
Contact Lens Wear
Advise patients not to wear contact lenses when using LOTEMAX.
Risk of Secondary Infection
Advise the patient to consult a physician if pain develops, redness, itching or inflammation becomes aggravated.
Distributed by:
Bausch & Lomb Americas Inc.
Bridgewater, NJ 08807 USA
Manufactured by:
Bausch & Lomb Incorporated
Tampa, FL 33637 USA
LOTEMAX is a trademark of Bausch & Lomb Incorporated or its affiliates.
© 2023 Bausch & Lomb Incorporated or its affiliates
9269204 Folded
9269104 Flat
- PACKAGE/LABEL PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
LOTEMAX
loteprednol etabonate gelProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:24208-503 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LOTEPREDNOL ETABONATE (UNII: YEH1EZ96K6) (LOTEPREDNOL - UNII:Z8CBU6KR16) LOTEPREDNOL ETABONATE 5 mg in 1 g Inactive Ingredients Ingredient Name Strength BORIC ACID (UNII: R57ZHV85D4) EDETATE DISODIUM (UNII: 7FLD91C86K) GLYCERIN (UNII: PDC6A3C0OX) POLYCARBOPHIL (UNII: W25LM17A4W) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SODIUM CHLORIDE (UNII: 451W47IQ8X) TYLOXAPOL (UNII: Y27PUL9H56) WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:24208-503-07 1 in 1 CARTON 10/12/2012 1 5 g in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:24208-503-48 1 in 1 CARTON 10/12/2012 2 0.5 g in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:24208-503-96 1 in 1 CARTON 10/12/2012 3 5 g in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA202872 10/12/2012 Labeler - Bausch & Lomb Incorporated (196603781) Establishment Name Address ID/FEI Business Operations Bausch & Lomb Incorporated 079587625 MANUFACTURE(24208-503) , LABEL(24208-503) , PACK(24208-503)