Label: POVIDONE-IODINE GEL SWABSTICK- povidone iodine gel
- NDC Code(s): 65517-0036-1
- Packager: Dukal LLC
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated October 13, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Dukal Povidone-Iodine Gel Swabstick Non-Sterile
- Active Ingredient
- Use
- Warnings
- Directions
- Other Information
- Inactive Ingredients
-
Dukal Povidone-Iodine Gel Swabstick
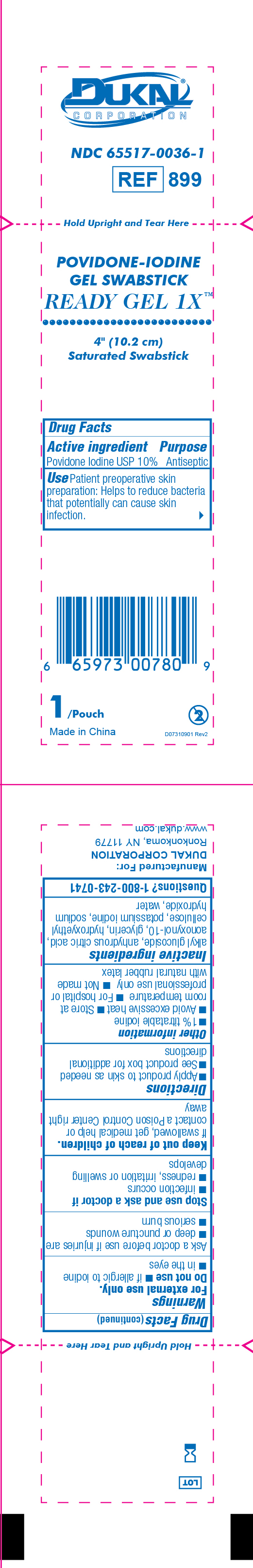 DUKAL™
DUKAL™
CORPORATIONNDC 65517-0036-1
REF 899
Hold Upright and Tear Here
POVIDONE-IODINE
Gel Swabstick
Ready Gel 1X™
4" (10.2cm)
Saturated Swabstick
Drug Facts
Active ingredients Purpose
Povidone Iodine USP 10% AntisepticUse Patient preoperative skin preparation: Helps to
reduce bacterial that potentially can cause skin infection
1/Pouch 2
Made in China D07310901 Rev2
-
INGREDIENTS AND APPEARANCE
POVIDONE-IODINE GEL SWABSTICK
povidone iodine gelProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:65517-0036 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 10 mg in 1 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) HYDROXYETHYL CELLULOSE (5000 CPS AT 1%) (UNII: X70SE62ZAR) POTASSIUM IODIDE (UNII: 1C4QK22F9J) GLYCERIN (UNII: PDC6A3C0OX) ANHYDROUS CITRIC ACID (UNII: XF417D3PSL) SODIUM HYDROXIDE (UNII: 55X04QC32I) NONOXYNOL-10 (UNII: K7O76887AP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:65517-0036-1 2.3 g in 1 POUCH; Type 0: Not a Combination Product 12/02/2019 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 12/02/2019 Labeler - Dukal LLC (791014871)

