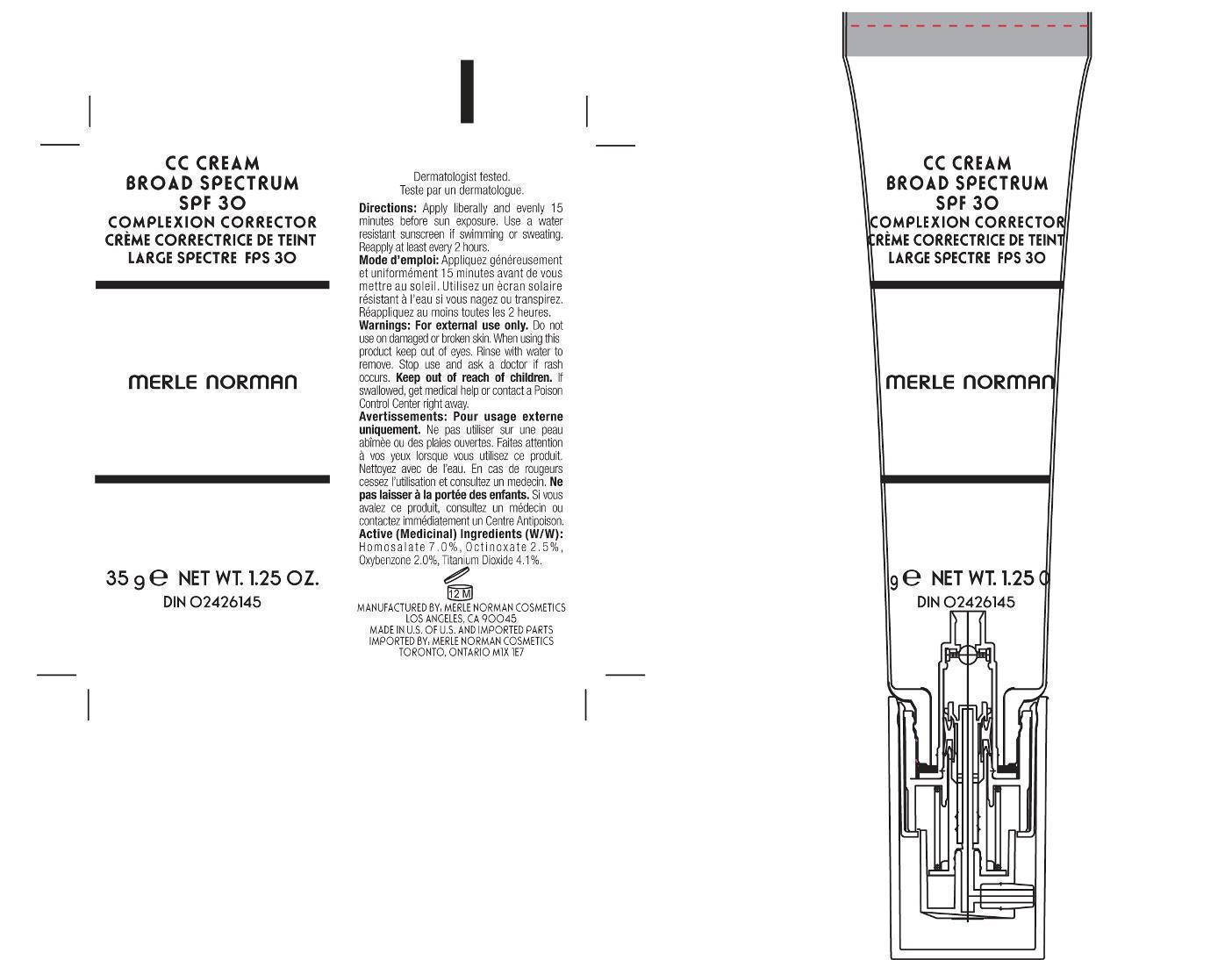
Label: CC CREAM BROAD SPECTRUM SPF 30 MERLE NORMAN- homosalate, octinoxate, oxybenzone, titanium dioxide cream
- NDC Code(s): 57627-170-01, 57627-170-02
- Packager: Merle Norman Cosmetics
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 22, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
ACTIVE INGREDIENT
Active (medicinal) Ingredients (w/w) Purpose
Homosalate 7.0% SunscreenOctinoxate 2.5% Sunscreen
Oxybenzone 2.0% Sunscreen
Titanium Dioxide 4.1% Sunscreen
Uses
• helps prevent sunburn
• if used as directed withother sun protection measures (see Directions), decreases the risk of skin cancer and early skin aging caused by the sun
Utilisations
• aide à prévenir les coups de soleil
• amoindrit le risque de cancer et de vieillissement prématuré de la peau causés par le soleil lorsque utilisé comme indiqué en conjonction avec d’autres mesures de protection solaire (voir Mode d’emploi).
Keep out of reach of children
Stop use and ask a doctor if rash occurs. En cas de rougeurs cessez l'utilisationet consultez un medecin.
Warnings
For external use only
Do not use on damaged or broken skin When using this product
keep out of eyes. Rinse with water to remove.
If swallowed, get medical help or contact a Poison Control Center right away.
Avertissements
À usage externe seulement
Ne pas utilisersur une peau abîmée ou des plaies ouvertes
Faites attention à vos yeux
lorsque vous utilisez ce produit. Nettoyez avec de l’eau.
Ne pas laisser à la portée des enfants.
Si vous avalez ce produit, consultez un médecin ou contactez immédiatement un centre antipoison.
Drug Facts
Directions
• For sunscreen use:
• apply liberally and evenly 15 minutes before sun exposure
• use a water resistant sunscreen if swimming or sweating
• reapply at least every 2 hours
•Sun Protection Measures.
Spending time in the sun increases your risk of skin cancer and early skin aging.
To decrease this risk, regularly use a sunscreen with a Broad Spectrum SPF value of 15 or higher and other sun protection measures including:
• limit time in the sun, especially from 10 a.m.-2 p.m.
• wear long-sleeved shirts, pants, hats, and sunglasses
• children under 6 months: Ask a doctor
Mode d’emploi
• Pour vous protéger de soleil:
• appliquez généreusement et uniformément 15 minutes avant de vous mettre au soleil
• utilisez un écran solaire résistant a l’eau si vous nagez ou transpirez
• réappliquez au moins toutes les 2 heures
• Mesures de protection contre le soleil.
L’exposition au soleil accroît le risque de cancer et de vieillissement prématuré de la peau. Pour amoindrir ce risque, utilisez
régulièrement un écran solaire à ample protection FPS 15 ou plus et observez d’autres mesures de protection contre le soleil
telles que:
• limiter le temps d’exposition au soleil, surtout entre 10 heures et 14 heures
• porter des chemises à manches longues, des pantalons, des chapeaux et des lunettes de soleil
• pour les enfants de moins de 6 mois: Consulter un médecin
Inactive (non-medicinal)ingredients
Water (Aqua), Methy l Trimethicone, Propylene Glycol Dicaprylate/Dicaprate, Isododecane, C12-15 Alkyl Benzoate, Cetyl PEG/PPG-10/1 Dimethicone, Glycerin, HDI/Trimethylol Hexyllactone Crosspolymer, Silica, Vinyl Dimethicone/Methicone Silsesquioxane Crosspolymer, Dimethicone, Magnesium Sulfate, Lauroyl Lysine, Tocopheryl Acetate, CitrullusLanatus (Watermelon) Fruit Extract, Lens Esculenta (Lentil) Fruit Extract, Pyrus Malus(Apple) Fruit Extract, Hippophae Rhamnoides Fruit Extract,Portulaca Oleracea Extract,Palmitoyl Tripeptide-38, Sodium PCA, Sodium Lactate,Sodium Ascorbyl Glucoside, Sodium Myristoyl Glutamate,Maltodextrin, Sorbitan Sesquioleate, Aluminum Hydroxide (CI 77002), Stearic Acid, Cyclopentasiloxane, Caprylyl Glycol, Zinc Oxide (CI 77947), Butylene Glycol, Ethylhexylglycerin, Acrylates/ Dimethicone Copolymer, Dimethicone/PEG-10/15 Crosspolymer, Triethoxycaprylylsilane, Sorbic Acid, Trisodium EDTA, Hydroxypropyl Cyclodextrin, Potassium Sorbate, Sodium Benzoate, Disteardimonium Hectorite, Propylene Carbonate, Phenoxyethanol, Methylparaben, Propylparaben, Ethylparaben,
May Contain/ Peut Contenir (+/-), Iron Oxides (CI 77489, CI 77491, CI 77492, CI 77499), Titanium Dioxide (CI 77891).
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
CC CREAM BROAD SPECTRUM SPF 30 MERLE NORMAN
homosalate, octinoxate, oxybenzone, titanium dioxide creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:57627-170 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength HOMOSALATE (UNII: V06SV4M95S) (HOMOSALATE - UNII:V06SV4M95S) HOMOSALATE 7 g in 100 g OCTINOXATE (UNII: 4Y5P7MUD51) (OCTINOXATE - UNII:4Y5P7MUD51) OCTINOXATE 2.5 g in 100 g OXYBENZONE (UNII: 95OOS7VE0Y) (OXYBENZONE - UNII:95OOS7VE0Y) OXYBENZONE 2.0 g in 100 g TITANIUM DIOXIDE (UNII: 15FIX9V2JP) (TITANIUM DIOXIDE - UNII:15FIX9V2JP) TITANIUM DIOXIDE 4.1 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) METHYL TRIMETHICONE (UNII: S73ZQI0GXM) PROPYLENE GLYCOL DICAPRYLATE/DICAPRATE (UNII: O4446S9CRA) ISODODECANE (UNII: A8289P68Y2) ALKYL (C12-15) BENZOATE (UNII: A9EJ3J61HQ) CETYL PEG/PPG-10/1 DIMETHICONE (HLB 2) (UNII: V2W71V8T0X) GLYCERIN (UNII: PDC6A3C0OX) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) DIMETHICONE (UNII: 92RU3N3Y1O) MAGNESIUM SULFATE, UNSPECIFIED FORM (UNII: DE08037SAB) LAUROYL LYSINE (UNII: 113171Q70B) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) WATERMELON (UNII: 231473QB6R) LENS CULINARIS FRUIT (UNII: ZYZ076G9JH) APPLE (UNII: B423VGH5S9) HIPPOPHAE RHAMNOIDES FRUIT (UNII: AVL0R9111T) PURSLANE (UNII: M6S840WXG5) SODIUM PYRROLIDONE CARBOXYLATE (UNII: 469OTG57A2) SODIUM LACTATE (UNII: TU7HW0W0QT) SODIUM MYRISTOYL GLUTAMATE (UNII: AYU7QD893W) MALTODEXTRIN (UNII: 7CVR7L4A2D) SORBITAN SESQUIOLEATE (UNII: 0W8RRI5W5A) ALUMINUM HYDRIDE (UNII: KZJ3T010RQ) STEARIC ACID (UNII: 4ELV7Z65AP) CYCLOMETHICONE 5 (UNII: 0THT5PCI0R) CAPRYLYL GLYCOL (UNII: 00YIU5438U) ZINC OXIDE (UNII: SOI2LOH54Z) BUTYLENE GLYCOL (UNII: 3XUS85K0RA) ETHYLHEXYLGLYCERIN (UNII: 147D247K3P) TRIETHOXYCAPRYLYLSILANE (UNII: LDC331P08E) SORBIC ACID (UNII: X045WJ989B) EDETATE TRISODIUM (UNII: 420IP921MB) POTASSIUM SORBATE (UNII: 1VPU26JZZ4) SODIUM BENZOATE (UNII: OJ245FE5EU) DISTEARDIMONIUM HECTORITE (UNII: X687XDK09L) PROPYLENE CARBONATE (UNII: 8D08K3S51E) PHENOXYETHANOL (UNII: HIE492ZZ3T) METHYLPARABEN (UNII: A2I8C7HI9T) ETHYLPARABEN (UNII: 14255EXE39) FERROUS OXIDE (UNII: G7036X8B5H) FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:57627-170-02 1 in 1 CARTON 06/19/2014 1 NDC:57627-170-01 35 g in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part352 06/19/2014 Labeler - Merle Norman Cosmetics (008479388) Registrant - Merle Norman Cosmetics (008479388) Establishment Name Address ID/FEI Business Operations Merle Norman Cosmetics 008479388 manufacture(57627-170)