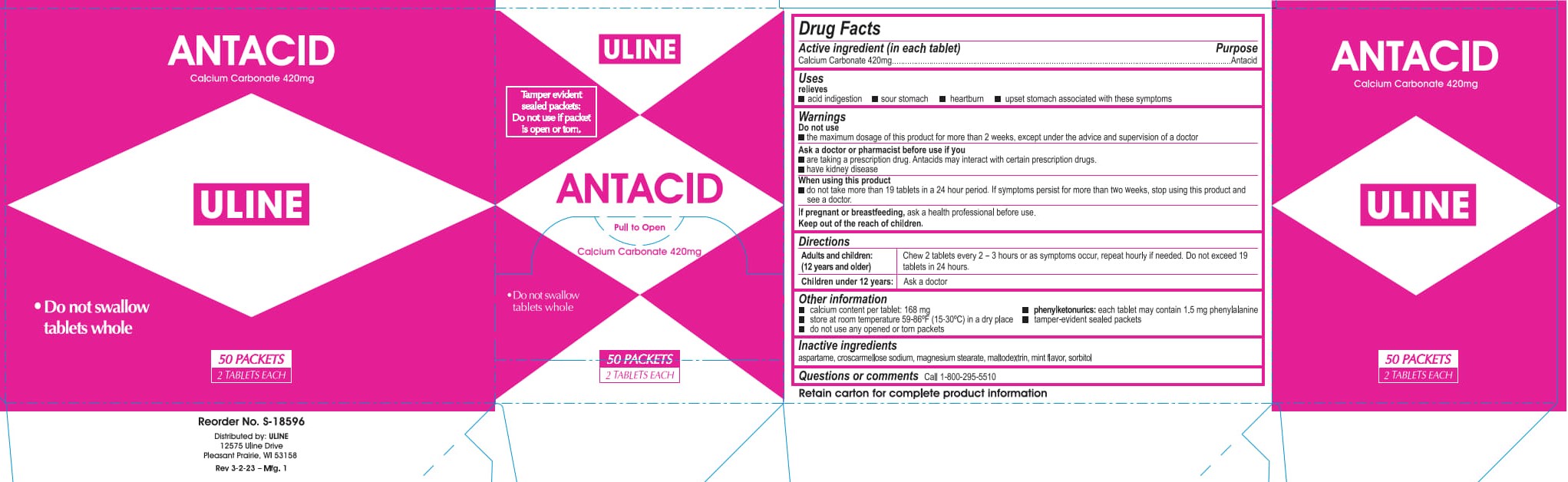
Label: ULINE ANTACID- calcium carbonate tablet, chewable
- NDC Code(s): 69790-089-33, 69790-820-33
- Packager: Uline
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated January 26, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- PURPOSE
- INDICATIONS & USAGE
- WARNINGS
- DO NOT USE
- ASK DOCTOR/PHARMACIST
- WHEN USING
- PREGNANCY OR BREAST FEEDING
- KEEP OUT OF REACH OF CHILDREN
- DOSAGE & ADMINISTRATION
- OTHER SAFETY INFORMATION
- INACTIVE INGREDIENT
- QUESTIONS
- PRINCIPAL DISPLAY PANEL
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ULINE ANTACID
calcium carbonate tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69790-089 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB, CARBONATE ION - UNII:7UJQ5OPE7D) CALCIUM CARBONATE 420 mg Inactive Ingredients Ingredient Name Strength MALTODEXTRIN (UNII: 7CVR7L4A2D) MAGNESIUM STEARATE (UNII: 70097M6I30) SUCROSE (UNII: C151H8M554) ACACIA (UNII: 5C5403N26O) Product Characteristics Color white Score no score Shape ROUND Size 12mm Flavor MINT Imprint Code FR;8 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69790-089-33 50 in 1 BOX 10/07/2019 03/01/2021 1 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M001 10/07/2019 03/01/2021 ULINE ANTACID
calcium carbonate tablet, chewableProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:69790-820 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength CALCIUM CARBONATE (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB, CARBONATE ION - UNII:7UJQ5OPE7D) CALCIUM CARBONATE 420 mg Inactive Ingredients Ingredient Name Strength MAGNESIUM STEARATE (UNII: 70097M6I30) MALTODEXTRIN (UNII: 7CVR7L4A2D) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) SORBITOL (UNII: 506T60A25R) Product Characteristics Color white Score no score Shape ROUND Size 11mm Flavor Imprint Code AZ;036 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:69790-820-33 50 in 1 BOX 10/07/2019 1 2 in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M001 10/07/2019 Labeler - Uline (039612668) Registrant - Unifirst First Aid Corporation (832947092) Establishment Name Address ID/FEI Business Operations Prestige Packaging 080667761 pack(69790-089, 69790-820) Establishment Name Address ID/FEI Business Operations Medique Products 086911794 pack(69790-820, 69790-089)