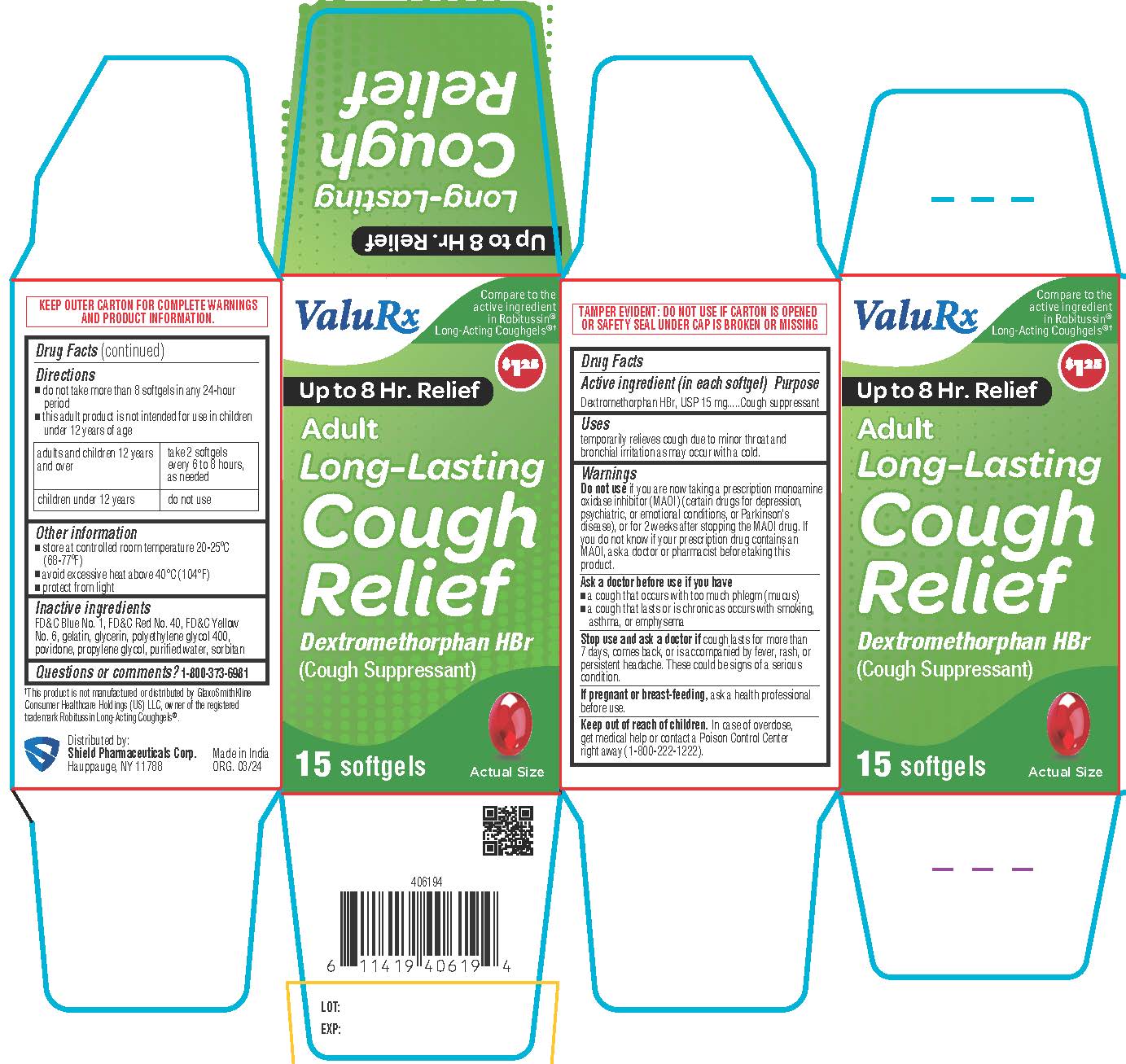
Label: ADULT LONG LASTING COUGH RELIEF- dextromethorphan hbr capsule, liquid filled
- NDC Code(s): 83059-0027-1
- Packager: Shield Pharmaceuticals Corp
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
Drug Label Information
Updated April 8, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredient (in each softgel)
- Purpose
- Uses
-
Warnings
Do not use
if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.Ask a doctor before use if you have
• a cough that occurs with too much phlegm (mucus)
• a cough that lasts or is chronic as occurs with smoking, asthma, or emphysema - Directions
- Other information
- Questions or comments?
- Inactive ingredients
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL
-
INGREDIENTS AND APPEARANCE
ADULT LONG LASTING COUGH RELIEF
dextromethorphan hbr capsule, liquid filledProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:83059-0027 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 15 mg Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) FD&C YELLOW NO. 6 (UNII: H77VEI93A8) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POVIDONE (UNII: FZ989GH94E) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) SORBITAN (UNII: 6O92ICV9RU) WATER (UNII: 059QF0KO0R) Product Characteristics Color pink Score no score Shape CAPSULE Size 10mm Flavor Imprint Code SD29 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:83059-0027-1 1 in 1 CARTON 04/08/2024 1 15 in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M012 04/08/2024 Labeler - Shield Pharmaceuticals Corp (118724924)