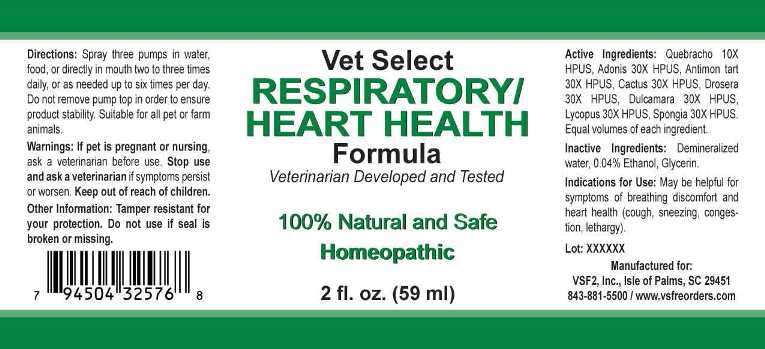
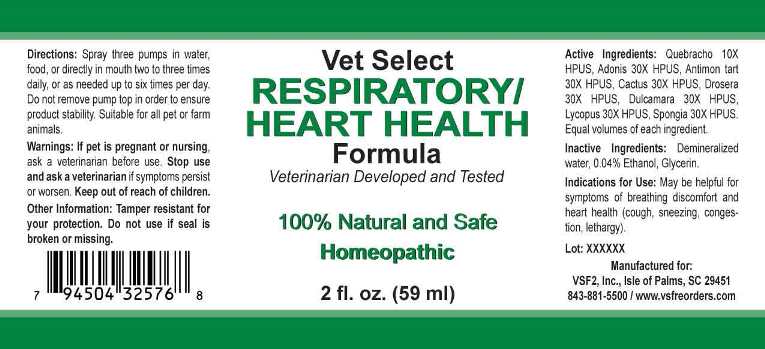
Label: RESPIRATORY/HEART HEALTH (quebracho, adonis vernalis, antimonium tartaricum, cactus grandiflorus, drosera- rotundifolia, dulcamara, lycopus virginicus, spongia tosta liquid
- NDC Code(s): 86100-0004-1
- Packager: Vsf2, Inc.
- Category: OTC ANIMAL DRUG LABEL
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated October 26, 2022
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- ACTIVE INGREDIENTS:
- INDICATIONS FOR USE:
- WARNINGS:
- KEEP OUT OF THE REACH OF CHILDREN:
- DIRECTIONS:
- INDICATIONS FOR USE:
- INACTIVE INGREDIENTS:
- QUESTIONS:
- PACKAGE LABEL DISPLAY:
-
INGREDIENTS AND APPEARANCE
RESPIRATORY/HEART HEALTH
quebracho, adonis vernalis, antimonium tartaricum, cactus grandiflorus, drosera (rotundifolia), dulcamara, lycopus virginicus, spongia tosta liquidProduct Information Product Type OTC ANIMAL DRUG Item Code (Source) NDC:86100-0004 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ASPIDOSPERMA QUEBRACHO-BLANCO BARK (UNII: 52B1340190) (ASPIDOSPERMA QUEBRACHO-BLANCO BARK - UNII:52B1340190) ASPIDOSPERMA QUEBRACHO-BLANCO BARK 10 [hp_X] in 1 mL ADONIS VERNALIS (UNII: DX3ZNI25WK) (ADONIS VERNALIS - UNII:DX3ZNI25WK) ADONIS VERNALIS 30 [hp_X] in 1 mL ANTIMONY POTASSIUM TARTRATE (UNII: DL6OZ476V3) (ANTIMONY CATION (3+) - UNII:069647RPT5) ANTIMONY POTASSIUM TARTRATE 30 [hp_X] in 1 mL SELENICEREUS GRANDIFLORUS STEM (UNII: 7114SV0MYK) (SELENICEREUS GRANDIFLORUS STEM - UNII:7114SV0MYK) SELENICEREUS GRANDIFLORUS STEM 30 [hp_X] in 1 mL DROSERA ROTUNDIFOLIA (UNII: QR44N9XPJQ) (DROSERA ROTUNDIFOLIA - UNII:QR44N9XPJQ) DROSERA ROTUNDIFOLIA 30 [hp_X] in 1 mL SOLANUM DULCAMARA TOP (UNII: KPS1B1162N) (SOLANUM DULCAMARA TOP - UNII:KPS1B1162N) SOLANUM DULCAMARA TOP 30 [hp_X] in 1 mL LYCOPUS VIRGINICUS (UNII: TWH5125Q6F) (LYCOPUS VIRGINICUS - UNII:TWH5125Q6F) LYCOPUS VIRGINICUS 30 [hp_X] in 1 mL SPONGIA OFFICINALIS SKELETON, ROASTED (UNII: 1PIP394IID) (SPONGIA OFFICINALIS SKELETON, ROASTED - UNII:1PIP394IID) SPONGIA OFFICINALIS SKELETON, ROASTED 30 [hp_X] in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) ALCOHOL (UNII: 3K9958V90M) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:86100-0004-1 59 mL in 1 BOTTLE, SPRAY Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved homeopathic 01/31/2019 Labeler - Vsf2, Inc. (034543894) Registrant - Apotheca Company (844330915) Establishment Name Address ID/FEI Business Operations Apotheca Company 844330915 manufacture, api manufacture, label, pack